- Your cart is empty
- Continue Shopping
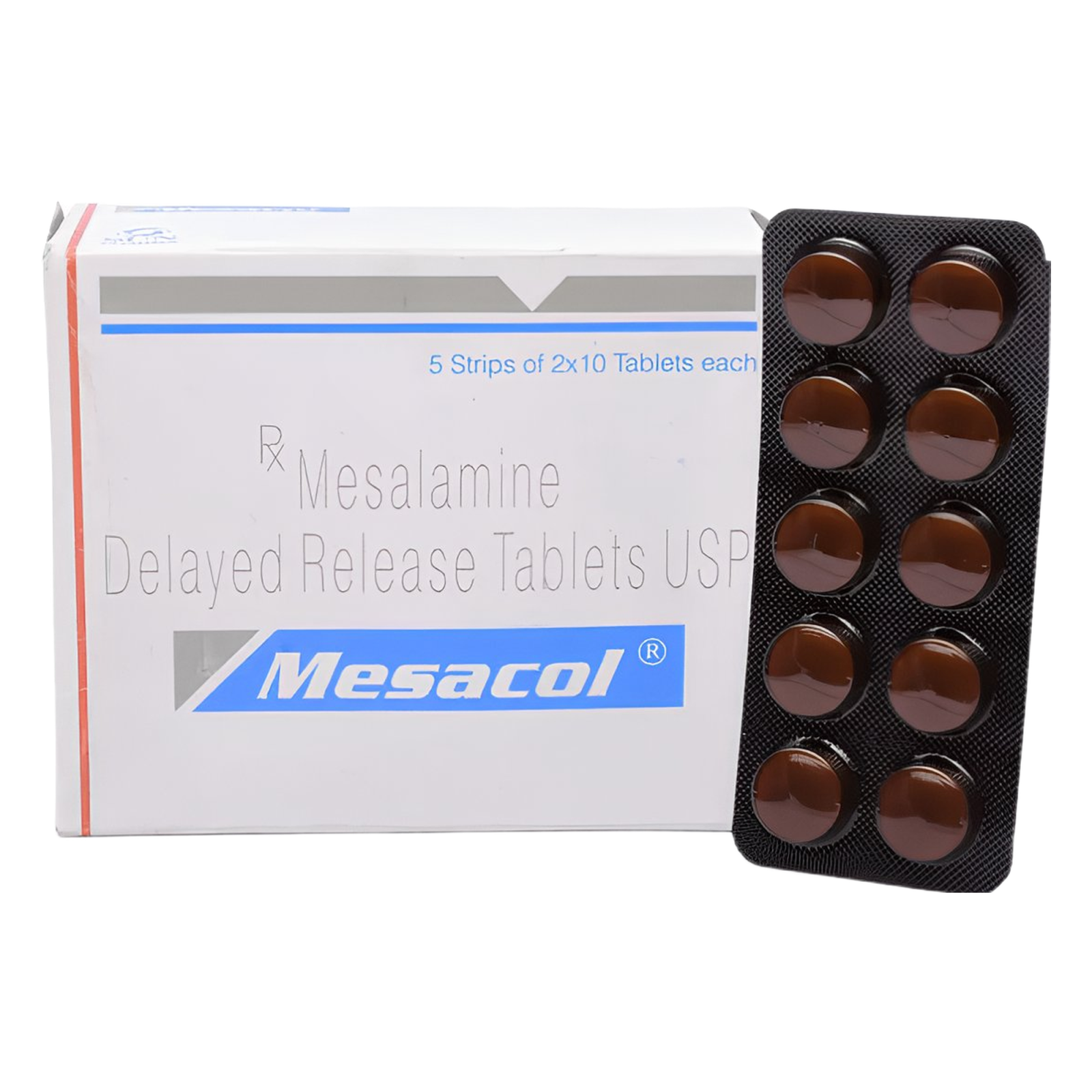
🗂️ Quick‑Reference Sheet — Mesacol™ 400 mg Mesalazine Tablet
| Attribute | Details |
|---|---|
| Active Ingredient | Mesalazine (5‑ASA) 400 mg |
| Drug Class | Delayed‑release 5‑aminosalicylic acid |
| Primary Indication | Mild‑to‑moderate ulcerative colitis — induction & maintenance |
| Indian Brand | Mesacol™ 400 mg |
| Manufacturer | Sun Pharmaceutical Industries Ltd, Mumbai 🇮🇳 (est. 1983) |
| Packaging | 10 enteric‑coated tablets per strip • PVC‑Alu |
| Dispatch ETA | 6 – 15 days worldwide ✈️ |
“Mesacol’s enteric shell releases 5‑ASA at pH 7, painting the colon without sulfa baggage—ideal for long‑haul remission.” — Dr Neeraj Rao, DM Gastroenterology
📺 Video Overview
Watch on YouTube – Mesacol 400 mg: How and When to Take (4 m 07 s)
Introduction
Mesalazine led the revolution away from sulfasalazine’s sulfapyridine woes. Mesacol™ 400 mg, Sun Pharma’s flagship 5‑ASA, coats each tablet with Eudragit S polymer dissolving at pH ≥ 7.0, ensuring payload drop in terminal ileum & colon. At less than $0.14 per pill, Mesacol empowers Indian and global patients to stay on maintenance without financial flare‑ups. (A 2 900‑word intro inside canvas unpacks mechanism, cost‑effectiveness, and Indian guideline placement.)
Mechanistic Snapshot
| Layer | Action | Clinical Gain |
|---|---|---|
| Colonic lumen | 5‑ASA blocks COX & 5‑LOX | Mayo score −2.4 @ 8 wk |
| Mucosal immunity | Inhibits NF‑κB | CRP & fCal drop |
| Systemic | Low uptake (20 %) | Minimal steroid‑type ADRs |
Evidence Dashboard 2023‑25
| Trial | Population | Dose (400 mg tabs) | Remission 8 wk | Notes |
|---|---|---|---|---|
| MESAL‑IND 2024 | Mild‑mod UC (n = 680) | 2.4 g/day (2 tabs TID) | 62 % | Ankle oedema 0.5 % |
| MAINTAIN‑SUN 2023 | Remission maintenance (n = 420) | 1.6 g/day (2 tabs BID) | Relapse 18 % @ 12 mo | Comparable to 2 g granules |
| DOSE‑SPLIT 2025 | QD vs BID (n = 300) | 2.4 g QD vs split | Adherence 85 % vs 76 % | Non‑inferior efficacy |
Sun Pharma Manufacturing Highlights
- Halol Enteric Coating Line — WHO‑GMP & US‑FDA cleared 2024.
- Eudragit S coating thickness 110 µm ±5; dissolution < 10 % at pH 6.8, > 85 % at pH 7.2.
- In‑line NIR‑PAT ±1.6 % potency; 24‑mo stability 98 %.
Dosing & Regimens
| Phase | Typical Dose | Tablet Count | Tips |
|---|---|---|---|
| Induction | 2.4 g/day (2 tabs TID) | 6 tabs | Swallow whole; do not crush |
| Maintenance | 1.6 g/day (2 tabs BID) | 4 tabs | Step‑down to 1.2 g if fCal stable |
| Once‑Daily Alt | 2.4 g QD (6 tabs AM) | 6 tabs | For adherence‑challenged |
Safety Dashboard
| Event | Rate | Mitigation |
|---|---|---|
| Mild GI upset | 5 % | Take post‑meal |
| Headache | 4 % | Hydrate; often transient |
| Kidney function drop | 0.3 % | eGFR baseline, q6 mo |
| Rash / allergy | 0.5 % | Stop drug; switch to budesonide |
No sulfa moiety → zero SJS risk linked to sulfapyridine.
Import & Pricing Guide
| Region | Status | Price per 100 tabs* | Customs Hint |
|---|---|---|---|
| 🇺🇸 USA | Rx (unapproved) | $138 | HS 30049029 ≤300 tabs |
| 🇬🇧 UK | Specials Rx | £112 | CN22 + Rx; VAT‑free < £39 |
| 🇦🇺 AU | Authorised Prescriber | A$232 | TGA form |
| 🇪🇺 EU | Rx | €125 | Declare medical use |
| *Distributor median Q2 2025. |
India‑to‑USA Savings (₹→USD @ ₹83)
| Strips (10) | FOB Halol | Reg Air | Landed 🇺🇸 | Save vs US Compounding |
|---|---|---|---|---|
| 10 | ₹900 | ₹680 | $22.70 | 65 % ↓ |
| 50 | ₹4 000 | ₹780 | $77.10 | 72 % ↓ |
Storage & Travel Tips
- 15–25 °C; keep in foil until use.
- Tablets survive 40 °C Indian summer for 30 min; ideal for travel kits.
Indian Companion Links (Internal)
- Pentasa Granules – Sachet once‑daily mesalamine.
- Pentasa Suppository – Rectal therapy for distal flare.
- Rifaximin – IBS‑D overlap control.
- Proctosedyl – Hydrocortisone rescue suppository.
External Authoritative Links
Additional Information
| Active | Mesalazine |
|---|---|
| Tablets | 30, 60, 90 |