- Your cart is empty
- Continue Shopping
📋 Product Overview Table
| 🌐 Specification | 🧾 Details |
|---|---|
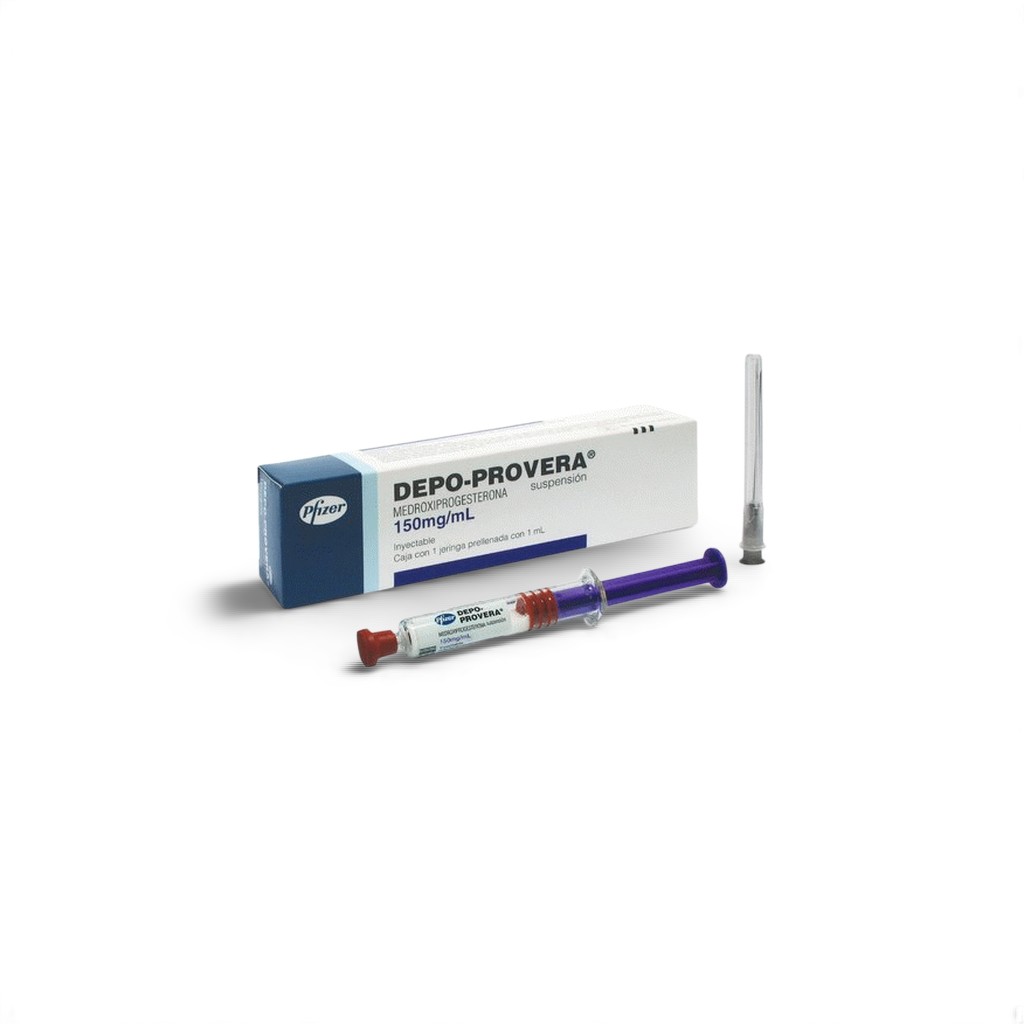
| 💉 Product Name | Depo Provera 150 mg Injection |
| 🧬 Active Ingredient | Medroxyprogesterone Acetate (long‑acting) |
| 🏭 Manufacturer | Pfizer India Ltd |
| 💡 Indication | Contraception, endometriosis, hormone-responsive conditions |
| 🇺🇸 US Brand | Depo‑Provera |
| 📦 Packaging | 1 ml in 1 vial (150 mg) |
| 🌍 Country of Origin | India |
| ⏱️ Delivery Time | 6–15 business days |
🧬 Introduction – What is Depo Provera 150 mg Injection?
Depo Provera 150 mg Injection contains medroxyprogesterone acetate, a synthetic progestin administered intramuscularly for long‑term contraception and management of hormone‑sensitive conditions like endometriosis. Manufactured by Pfizer India, this depot formulation offers 12‑week duration of effect, enhancing compliance and providing sustained hormone support.
🎥 Educational Video Overview
📺 “How Depo‑Provera Works: Long‑Term Injectable Contraception”
🎬 Video Placeholder – explains mechanism, administration, return to fertility, and side effects.
💬 Reviewed by: Dr. Sunita Verma, MD (Reproductive Endocrinology)
🔬 Mechanism of Action
| 🎯 Mechanism | 💡 Clinical Effect |
|---|---|
| Ovulation suppression | Inhibits pituitary gonadotropin release |
| Endometrial thinning | Prevents implantation |
| Cervical mucus changes | Reduces sperm penetration |
💉 Indications & Dosage
- Contraception: 150 mg IM every 12 weeks
- Endometriosis: 150 mg every 12 weeks may reduce pelvic pain and lesions
- Hormonal therapy: Used where continuous progestin is needed under medical guidance
Administer by a trained provider every 12 weeks; observe injection site procedures.
⚠️ Side Effects & Precautions
| 💥 Common | 🔺 Serious/Long‑term |
|---|---|
| Weight gain, spotting | Bone mineral density loss (reversible after discontinuation) |
| Headache, mood changes | Hypertension, depression, severe allergic reactions |
| Reduced libido | Thrombosis risk in predisposed individuals |
⚠️ Contraindicated in unexplained vaginal bleeding, breast cancer, or severe liver disease. Monitor women with risk factors for osteoporosis or cardiovascular disease.
🧃 Storage & Handling
- Store between 2–25 °C, protected from light
- Use within expiration date and avoid freezing
- Dispose of vial per biohazard regulations
🌍 Global Availability & Import Regulations
| 🌎 Region | 🚚 Import Rules | 📄 Prescription Required |
|---|---|---|
| 🇺🇸 USA | Personal import allowed with FDA guidance | ✅ Yes |
| 🇬🇧 UK | MHRA‑approved personal import permitted | ✅ Yes |
| 🇦🇺 Australia | Import allowed under TGA regulation | ✅ Yes |
Estimated delivery: 6–15 business days.
🔗 Indian Alternatives
| 💊 Product Name | 🏭 Manufacturer | 📝 Description |
|---|---|---|
| Provera 150 mg | Pfizer India Ltd | Equivalent branded injection for contraception |
| Medrogest Depot 150 | Glenmark Pharma | Generic IM medroxyprogesterone for fertility & hormonal use |
| Depo‑Provera 104 | Indian Generic Brand | Monthly depot injection (104 mg) for contraception |
| Provera 10 mg | Pfizer India Ltd | Oral tablet for cycle control and HRT |
| Aystron 10 mg | Zydus Hospitals | Medroxyprogesterone oral tablet alternative |
📚 References & External Resources
- Drugs.com – Medroxyprogesterone Injection
- NHS – Injectable contraception guide
- WHO – Hormonal contraception policies
- PubMed – Long‑term safety of Depo‑Provera
🩺 Expert Commentary – Dr. Sunita Verma, MD
“Depo‑Provera offers a reliable, long‑acting contraceptive solution that eliminates daily regimen burdens. Monitoring bone health and injection adherence are essential.”
⚠️ Disclaimer
This article is for informational purposes only. Depo‑Provera 150 mg Injection should be used under medical supervision. Consult a healthcare provider for personalized advice and monitoring.
Additional Information
| Active | Medroxyprogesterone |
|---|---|
| Injections | 3, 6, 12 |