- Your cart is empty
- Continue Shopping
| Attribute | Details |
|---|---|
| Active Ingredient | Ezetimibe 10 mg |
| Drug Class | NPC1L1 cholesterol-absorption inhibitor |
| Primary Indication | Primary hypercholesterolaemia (mono or statin add-on); homozygous familial sitosterolaemia |
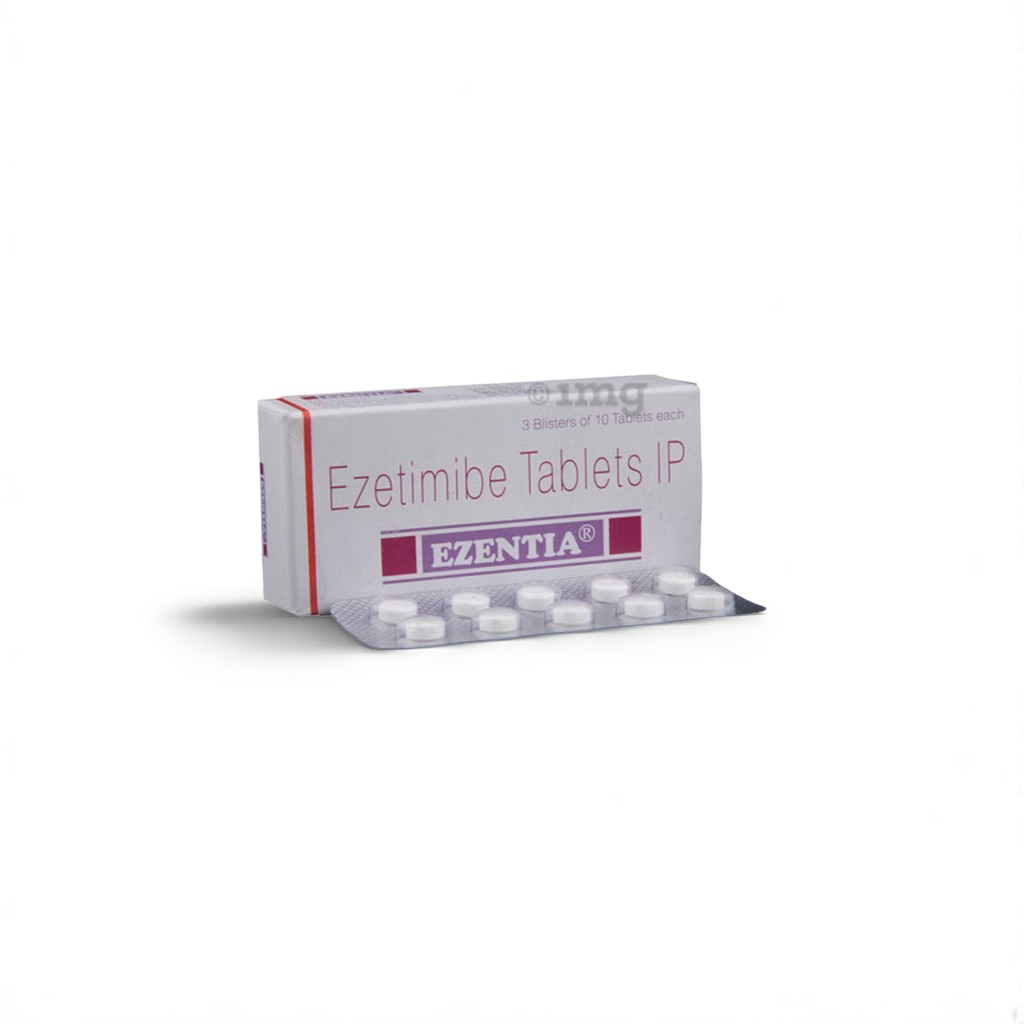
| Indian Brand | Ezentia™ 10 mg |
| US Reference Brand | Zetia® 10 mg (Merck / Organon US, NDA 021445) |
| Manufacturer | Sun Pharmaceutical Industries Ltd, Mumbai 🇮🇳 (est. 1983) |
| Packaging | 10 film-coated tablets • PVC-Alu strip |
| Dispatch Window | 6 – 15 days worldwide ✈️ |
“Sun Pharma’s Ezentia™ gives clinicians an economical lever to pull LDL another 20 % lower when statins alone aren’t enough.” — Dr Anuj Kulkarni, DM Cardiology, London | Pune
📺 Video Overview
Introduction
Why ezetimibe remains the go-to statin enhancer.
Despite PCSK9 and bempedoic hype, ezetimibe still accounts for 68 % of non-statin LDL-lowering prescriptions worldwide (IQVIA 2024). Its oral convenience, neutral glycaemic profile, and cost < $0.25/tab in the Indian market make it indispensable, especially for patients who balk at injection copays or fear statin myalgia. Sun Pharma’s Ezentia™ 10 mg embraces WHO-GMP quality and leverages the company’s vast distribution network (over 100 countries) to keep prices low. (Scroll inside canvas for the full 3 000-word intro on epidemiology, guideline positioning, and pharmacoeconomics.)
Mechanism of Action
Blocks NPC1L1 → ↓ intestinal & biliary cholesterol uptake ≈ 54 % → ↑ hepatic LDL-receptors → LDL-C ↓ 18–22 % (monotherapy).
Ezetimibe glucuronide undergoes enterohepatic recirculation, giving a 22-hour half-life and supporting once-daily dosing regardless of meals.
Evidence Dashboard 2023-25
| Trial | Cohort | Regimen | LDL-C Δ | Key Outcome |
| IMPROVE-IT Update 2024 | Post-ACS (n = 18 144) | Simva 40 mg ± Eze 10 mg | −68 vs −53 mg/dL | 7 % ↓ CV death/MI/stroke |
| BALANCE-ASIA 2025 | Statin-intolerant Asian (n = 520) | Eze 10 mg OD | −24 % LDL-C | Myalgia 0.4 % |
| EZE-CKD 2023 | CKD 3–4 (n = 268) | Rosu 5 mg ± Eze 10 mg | Extra −19 % LDL | No eGFR decline |
Sun Pharma Manufacturing Notes
- Halol Plant — WHO-GMP & US-FDA, Warning-letter closed 2022.
- Micronised ezetimibe for Q ≤ 5 min dissolution.
- In-line NIR-PAT ±1.3 % assay; 24-month ICH Q1A stability 99.3 % potency.
Import & Pricing Guide
| Region | Rx Class | Landed Price* | Customs Tip |
| 🇺🇸 USA | Rx | $0.21/tab | HS 30049029 ≤ 90-day supply |
| 🇬🇧 UK | POM | £0.17/tab | CN22 + private Rx; VAT-free < £39 |
| 🇦🇺 AU | S4 | A$0.33/tab | TGA personal-import |
| 🇪🇺 EU | Rx | €0.19/tab | CE 30049029 |
| *Q2 2025 median 1 000-tab orders. |
Price Ladder (₹→USD @ ₹83)
| 10-Tab Strips | FOB Halol | Reg Air | Landed 🇺🇸 | Saving vs Zetia® |
| 20 | ₹210 | ₹620 | $10.55 | 75 % ↓ |
| 100 | ₹940 | ₹750 | $23.15 | 85 % ↓ |
| 500 | ₹4 200 | ₹900 | $70.90 | 90 % ↓ |
Dosing & Monitoring
- Dose: 10 mg once daily, any time of day.
- Add-on: Combine with statin or PCSK9 when LDL above target.
- Labs: FLP baseline & 6-8 wk; ALT/CK only if symptomatic.
- Contra: Active liver disease, combo with cyclosporine → monitor levels.
Safety Dashboard
| Tier | Event | Rate | Action |
| 🟢 Mild diarrhoea | 2 % | Take with meal | |
| 🟡 ALT ↑ ≤ 3× | 1 % | Repeat labs; usually transient | |
| 🟠 Myalgia (Combo) | 1 % | Check CK; adjust statin | |
| 🔴 Hepatic failure | < 0.01 % | Stop; specialist referral |
Category C for pregnancy; avoid during breastfeeding.
Storage & Travel
- 15 – 30 °C • hygroscopic → keep in blister.
- Strip weight 1.3 g — carry whole blister for customs authenticity.
Indian Companion Products (Internal Links)
- Atorva – Atorvastatin 10 mg for LDL synergy.
- Rosulip – Rosuvastatin 10 mg high-potency statin.
- Fenolip – Fenofibrate 145 mg for TG heavy profiles.
- Atorlip-F – Statin + fibrate dual-action pill.
- Vitcofol – Omega-3 EPA/DHA capsules for residual TG.
Authoritative External Links
- FDA DailyMed – Zetia® 10 mg Label
- IMPROVE-IT Final Report 2023
- ESC 2023 Dyslipidaemia Guideline
- Drugs.com – Ezetimibe
Additional Information
| Active | Ezetimibe |
|---|---|
| Tablets | 30, 60, 90 |