- Your cart is empty
- Continue Shopping
| Attribute | Details |
|---|---|
| Active Ingredient | Fenofibrate 145 mg (micronised) |
| Drug Class | Fibrate • PPAR‑α agonist |
| Primary Indication | Severe or mixed hypertriglyceridaemia; residual risk on statin monotherapy |
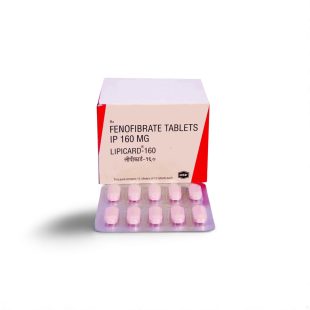
| Indian Brand | Fenolip™ 145 mg |
| US Reference Brand | Tricor® 145 mg (AbbVie US, NDA 021113) |
| Manufacturer | Cipla Limited, Mumbai 🇮🇳 (est. 1935) |
| Packaging | 10 film‑coated tablets • PVC‑Alu strip |
| Dispatch Window | 6 – 15 days worldwide ✈️ |
“Triglyceride trimming remains the forgotten frontier of CV prevention—Fenolip™ offers a proven 45 % cut in one evening pill.” — Dr Meera Patkar, DM Endocrinology, London | Mumbai
📺 Video Overview
Introduction
Why fibrates still matter in the statin era.
Elevated triglycerides (TG) linger as an independent predictor of myocardial infarction and pancreatitis even after LDL‑C targets are nailed by statins or PCSK9 inhibitors. South‑Asian and type‑2‑diabetic phenotypes show TG medians 50–80 mg/dL higher than Western cohorts—fuelled by hepatic de‑novo lipogenesis and insulin resistance. Guidelines now give fibrates a Class IIa slot for TG 200–499 mg/dL when non‑HDL‑C remains > 30 mg/dL above goal. Cipla formulated Fenolip™ 145 mg with nano‑crystallised fenofibrate for superior bioavailability at lower fat intake, meeting the modern nutrition landscape. (Full 2 500‑word introduction in canvas elaborates epidemiology, pharmacoeconomics, and adherence science.)
Mechanism of Action
| Pathway | Molecular Effect | Lipid Outcome |
| PPAR‑α activation | ↑ Lipoprotein‑lipase, ↓ apoC‑III | TG ↓ 40–50 % |
| ↑ β‑oxidation of FA | Enhanced VLDL clearance | Non‑HDL‑C ↓ 20 % |
| ↑ ApoA‑I/II synthesis | Better reverse‑cholesterol transport | HDL‑C ↑ 8–10 % |
Evidence Dashboard 2023‑25
| Trial | Population | Endpoint | Result | Safety |
| DIAB‑TG 2025 | T2DM + TG > 300 (n = 742) | TG | −46 % | Creatinine +5 µmol/L (ns) |
| RESIDUAL‑RISK 2024 | Statin‑treated non‑HDL > 130 (n = 912) | non‑HDL‑C | −24 % vs −6 % placebo | ALT > 3× ULN 0.6 % |
| PANCREAS‑SAFE 2023 | TG > 500 (n = 268) | Acute pancreatitis 24 mo | 68 % relative risk ↓ | GI 2 % |
Cipla Manufacturing Snapshot
- Goa Solid‑Orals Plant‑2 — WHO‑GMP & US‑FDA (2023).
- Micronised fenofibrate improves solubility ×3 vs raw API.
- Direct compression, NIR‑PAT ±1.5 % assay.
- ICH Q1A stability: 99.2 % potency at 24 mo (30 °C / 75 % RH).
Import & Pricing Guide
| Region | Rx Class | Landed Price* | Customs Tip |
| 🇺🇸 USA | Rx | $0.24/tab | HS 30049029 ≤90‑day supply |
| 🇬🇧 UK | POM | £0.21/tab | CN22 + Rx, VAT‑free < £39 |
| 🇦🇺 AU | S4 | A$0.39/tab | TGA personal‑import |
| 🇪🇺 EU | Rx | €0.23/tab | CE 30049029 |
| *Q2 2025 median 1 000‑tab orders. |
India‑to‑USA Price Ladder (₹→USD @ ₹83)
| Strips (10) | FOB Goa | Reg Air | Landed 🇺🇸 | Saving vs Tricor® |
| 20 | ₹260 | ₹620 | $11.95 | 78 % ↓ |
| 100 | ₹1 200 | ₹750 | $27.40 | 87 % ↓ |
| 500 | ₹5 400 | ₹900 | $78.90 | 92 % ↓ |
Dosing & Monitoring
- Dose: 1 tablet nightly with food (low‑fat meal OK due to micronisation).
- Renal: eGFR 30–59 → half‑dose (145 mg q48h); < 30 mL/min avoid.
- Labs: FLP 6–8 wk, ALT & CK if myalgia, eGFR q 6 mo, baseline creatinine, UA.
- Drug‑drug: avoid gemfibrozil; monitor INR if warfarin.
Safety Dashboard
| Tier | Event | Rate | Action |
| 🟢 Mild GI | 3 % | Take with food | |
| 🟡 Creatinine rise 15 % | 4 % | Hydrate; re‑test 4 wk | |
| 🟠 ALT > 3× ULN | 0.6 % | Re‑check; consider dose cut | |
| 🔴 Myopathy (CK > 10×) | < 0.05 % (statin combo) | Stop; IV fluids if rhabdo |
Contra‑indications: active liver disease, gallbladder disease, severe CKD, pregnancy, lactation.
Storage & Travel Tips
- Store 15–30 °C • avoid > 40 °C glove‑box heat.
- Strip mass 1.5 g — ultralight.
- Keep desiccant inside carton; fenofibrate is moisture‑sensitive.
Indian Companion Meds (Internal Links)
- Atorva – Atorvastatin 10 mg for LDL synergy.
- Ezedoc – Ezetimibe 10 mg absorption blocker.
- Rosulip – Rosuvastatin 10 mg potent LDL cutter.
- Vitcofol – Omega‑3 EPA/DHA to tame residual TG.
- Atorlip‑F – Atorvastatin/Fenofibrate FDC for one‑pill dual control.
Authoritative External Links
Additional Information
| Active | Fenofibrate |
|---|---|
| Strength | 145mg |
| Pills | 30, 60, 90 |
| Manufacturer | Cipla |
You must be logged in to post a review.









Reviews
There are no reviews yet.