- Your cart is empty
- Continue Shopping
✅ Active Ingredient Overview Table:
| 🔬 Active Ingredient (Generic Name): | Human Placental Extract (Hydrolyzed) |
|---|---|
| 💊 Indication: | Wound Healing, Tissue Regeneration, Chronic Ulcer Management |
| 🇺🇸 US Brand Equivalent: | No direct topical injection equivalent; regenerative injections by compounding pharmacies |
| 🏭 Manufacturer: | Albert David Limited (Founded: 1955) |
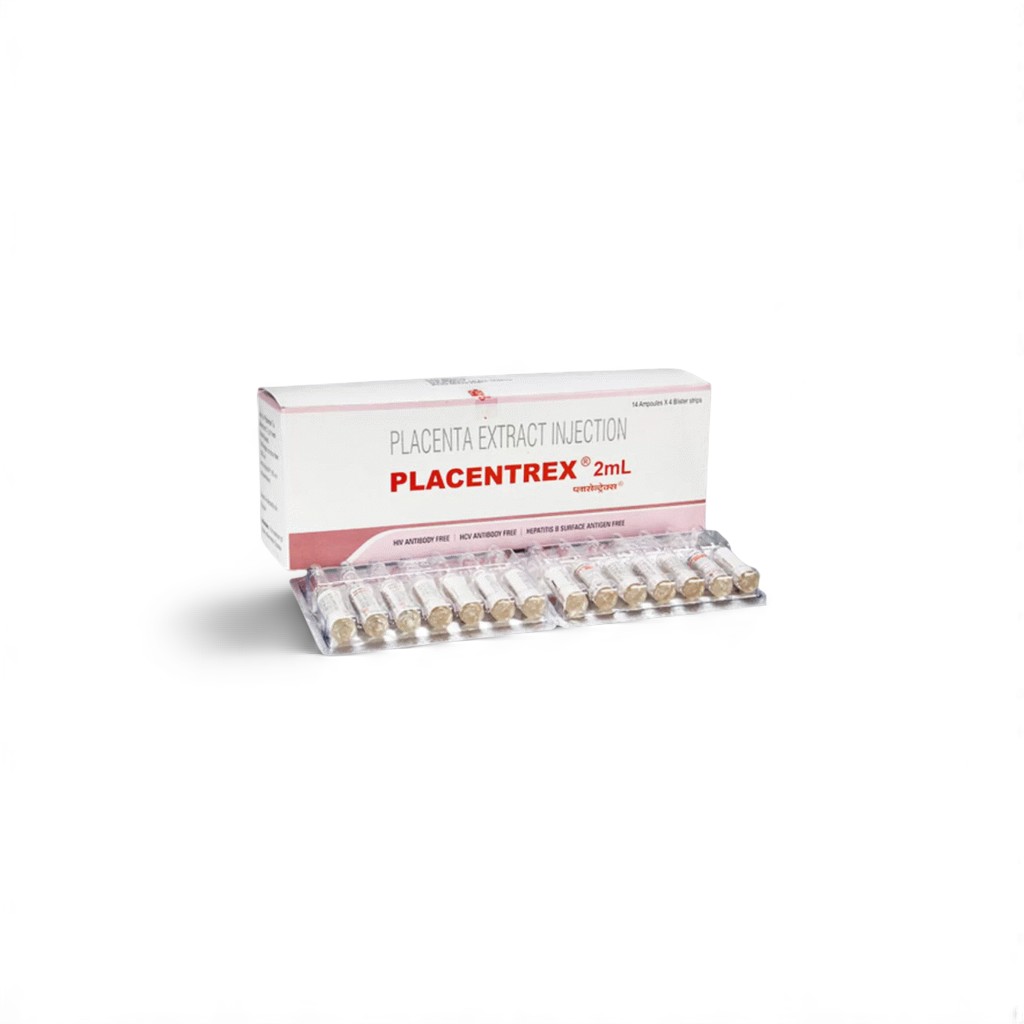
| 📦 Packaging: | 2 ml Vial (Lyophilized Powder) |
| 🧪 Strength: | 1000 IU placental extract equivalent per 2 ml after reconstitution |
| 🚚 Delivery Time: | 6–15 days |
🌿 What is Placentrex Injection 2 ml?
Placentrex Injection contains hydrolyzed human placental extract—rich in growth factors, peptides, and cytokines—formulated under nitrogen to maintain protein integrity. It is indicated for accelerating wound healing and promoting tissue regeneration in chronic non-healing wounds, diabetic foot ulcers, pressure sores, and surgical ulcers. By providing essential bioactive components directly into the target tissue, Placentrex Injection enhances fibroblast proliferation, angiogenesis, and extracellular matrix remodeling.
🧬 Pharmacology & Mechanism of Action
| Component | Mechanism |
|---|---|
| Human Placental Extract | • Contains growth factors (EGF, FGF, PDGF), cytokines, and amino acids → stimulates keratinocyte and fibroblast proliferation → accelerates re-epithelialization. |
| • Enhances collagen synthesis and neovascularization → supports formation of granulation tissue and wound closure. | |
| • Anti-inflammatory and antioxidant peptides reduce local inflammation and oxidative stress, creating a conducive environment for healing. | |
| Nitrogen Atmosphere | • Prevents oxidative degradation of labile proteins and peptides during storage → maintains biological activity without chemical preservatives. |
| Vehicle & Lyophilization | • Lyophilized powder ensures stability; reconstitution yields isotonic solution for direct injection into wound margins or perilesional tissue. |
💡 Indications & Administration Guidelines
| Condition | Administration Regimen |
|---|---|
| Chronic Non-Healing Wounds & Ulcers | • Dose: 2 ml reconstituted solution injected intradermally along wound margins and into base once weekly. |
| • Frequency: 1 injection per week for 4–6 weeks or until wound granulation and epithelialization significantly progress. | |
| Diabetic Foot Ulcers | • Prepare Wound: Debride necrotic tissue; cleanse area thoroughly. |
| • Injection: Inject 2 ml around ulcer periphery and subcutaneously into granulation tissue once weekly. | |
| • Adjunct: Combine with offloading techniques and glycemic control measures; continue until complete closure or plateau in healing. | |
| Pressure Sores & Bedsores | • Debridement & Cleaning: Remove slough and debris; maintain sterile field. |
| • Injection: 2 ml into periwound skin and subcutaneous tissue once weekly. | |
| • Duration: Continue for 4–8 weeks depending on wound grade and patient comorbidities; reassess weekly. | |
| Surgical & Traumatic Ulcers | • Timing: Administer first dose once wound is clean post-debridement (usually 3–5 days after injury/surgery). |
| • Injection: 2 ml intradermally around suture line or wound edges once weekly until sutures removed or wound closure is evident. |
- Preparation: Reconstitute lyophilized vial with 2 ml sterile normal saline; mix gently to avoid foaming. Use immediately after reconstitution.
- Technique: Use 26–27G needle to inject small aliquots (0.2–0.5 ml) at multiple points along wound edges and into granulation tissue. Maintain aseptic technique.
- Precautions: Do not inject into infected tissue; ensure infection is controlled with appropriate antibiotics before use. Avoid intravascular injection.
⚠️ Warnings & Side Effects
Common (Local):
- Mild, transient burning or stinging during injection.
- Minor erythema or edema at injection site lasting 1–2 days.
- Slight hematoma formation if small vessels punctured.
Less Common:
- Localized allergic reaction (pruritus, urticarial rash) → discontinue if occurs.
- Injection site infection if aseptic technique not maintained.
Serious (Rare):
- Systemic hypersensitivity or anaphylaxis → immediate discontinuation and emergency management.
- Transmission risk of blood-borne pathogens if source material compromised → however, human placental extracts sourced with strict screening minimize risk.
Precautions:
- Hypersensitivity: Contraindicated in patients with known allergy to placental proteins or excipients.
- Infection Control: Do not use on actively infected wounds unless infection is adequately treated.
- Pregnancy & Lactation: Not indicated for pregnant patients; no data on breastfeeding safety.
- Veteran Use: No pediatric safety established; use in adults only.
- Monitoring: Observe for local reactions; adjust or discontinue if significant adverse events occur.
🏥 Storage Instructions
Store Placentrex Injection vials at 2–8°C (refrigerate). Protect from light. Do not freeze. Use reconstituted solution immediately; discard any unused portion. Shelf life of lyophilized vials: 36 months from manufacturing date. Keep out of reach of children.
🌐 Availability & Purchasing
- United States: No FDA-approved human placental extract injections; available via compounding pharmacies or under physician special access.
- United Kingdom & EU: Prescription-only; specialized regenerative medicine clinics may stock similar products.
- Australia: TGA approval required; import under Special Access Scheme with prescription.
Importing Placentrex from India:
Purchasing Placentrex Injection from India offers cost advantages. Ensure compliance with local import regulations, maintain a valid prescription, and verify authenticity via batch certification to avoid counterfeit products.
🔗 Authoritative Resources
🔗 Injectable Therapies (Indian Brands)
- Placentrex Gel – Human Placental Extract/Nitrogen
🎥 Educational Video on Regenerative Wound Injections
👩⚕️ Expert Insight: Dr. Rohit Mehta, Wound Care Specialist
“Placentrex Injection provides direct delivery of growth factors and cytokines into the wound bed, expediting the proliferative phase of healing. Proper wound bed preparation and infection control are critical to maximize its regenerative potential.”
Additional Information
| Active | Human Placental Extract/Nitrogen |
|---|---|
| ampoule/s | 14, 28, 56 |