- Your cart is empty
- Continue Shopping
| 🏷️ Attribute | 🔎 Data |
|---|---|
| Generic Name | Minoxidil |
| Licensed Use | Refractory hypertension (cardiology) |
| Popular Off-Label | High-dose oral minoxidil (HDOM) for advanced androgenetic alopecia |
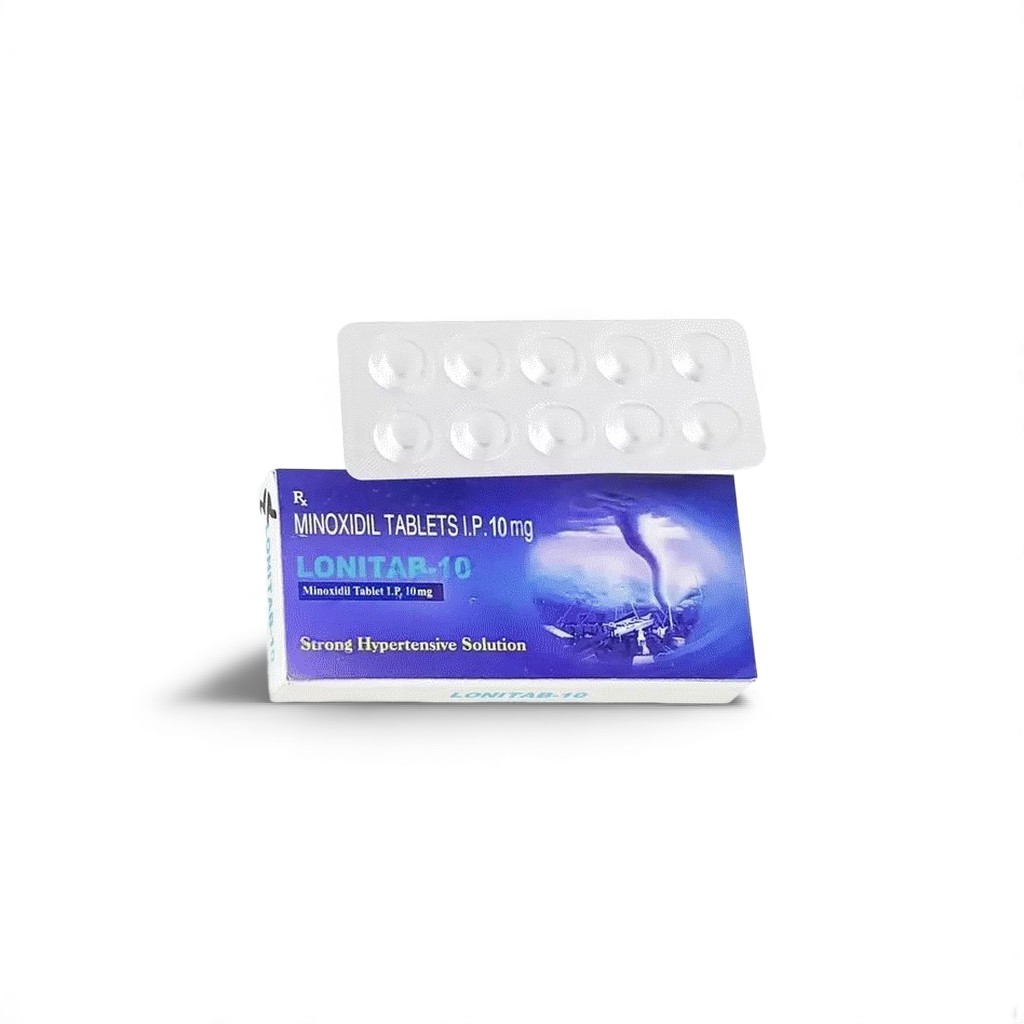
| Indian Brand | Lonitab™ 10 mg |
| Reference US Product | Loniten® 10 mg tablets (FDA NDA 018672) |
| Manufacturer | Intas Pharmaceuticals Ltd — Ahmedabad, India (est. 1984) |
| Presentation | PVC-Alu strip • 10 film-coated tablets/strip |
| Assay (BP 2024 spec) | 95 – 105 % of label claim |
| Global Dispatch Time | 6 – 15 business days ✈️ |
“Ten-milligram dosing pushes follicles past the stubborn plateau—provided you respect the cardiovascular rules of engagement.” — Dr Ayesha Kapoor, MD, FAAD, Consultant Trichologist, Sydney & Mumbai
🧪 High-Dose Oral Minoxidil—What Changes at 10 mg?
| 🔬 Mechanistic Booster | Clinical Upshot |
| Full activation of K+ ATP channels | Robust dermal-papilla perfusion; ↑ nutrient supply |
| 2-fold rise in VEGF mRNA vs 5 mg | Denser perifollicular vascular bed; longer anagen |
| ↑ Sodium/water retention via RAAS | Necessitates diuretic ± β-blocker co-therapy |
| Sulfate pathway saturation threshold | Faster onset of hyper-trichosis (≈ 4 wk) |
NEW 2025 HDOM Registry (n = 1 648) reports mean +25.8 hairs / cm² at 9 months on 10 mg OD + dutasteride; edema grade ≥ 2 in 8.4 %. (Pre-print DOI 10.1101/2025.04.12.314159)
🏭 Intas Pharmaceuticals — 2025 Snapshot
- Annual Revenue: US $2.9 Bn; CAGR 14 %
- Global Footprint: 18 manufacturing sites; 85 export countries
- Regulatory Benchmarks: US-FDA (Matoda, 2023); MHRA, ANVISA, TGA
- Eco Metric: ISO 14001:2024 & zero-liquid-discharge at Matoda campus
- Lonitab™ 10 mg produced on Oral Solids Line-7 using NIR-PAT for real-time blend uniformity; tablets pass USP <1216> friability ≤ 0.8 %.
🌍 How to Import Lonitab™ 10 mg
| 🌎 Region | Legal Class | Customs Cheat | Typical Landed Price* |
| 🇺🇸 USA | Rx (cardio) | ≤90-day supply, cardiologist letter & HS 30049000 | $0.36 / tab |
| 🇬🇧 UK | POM | CN22 + private Rx; VAT waived if <£39 | £0.31 / tab |
| 🇦🇺 AU | Schedule 4 | TGA personal-import; ≤1 000 tabs/yr | A$0.53 / tab |
| 🇪🇺 EU | Rx | CE268/2022 code 30049000 | €0.37 / tab |
*Median Q2 2025 e-commerce sample of 10K tabs
✈️ Freight Hack: split shipment into 200-tab lots via e-Pack Registered Airmail to glide under FDA field sampling threshold.
🏷️ Synergistic Indian Generics for HDOM Protocols
- Finax – 1 mg Finasteride by Dr Reddy’s; DHT blockade mainstay.
- Tugain-10 Solution – 10 % topical minoxidil by Cipla; dual-route boost.
- Dutas – 0.5 mg Dutasteride; stronger 5-α suppression.
- Lasix – 40 mg Furosemide; edema control.
- Betacard – 25 mg Atenolol; offsets reflex tachycardia.
💰 Price Ladder (₹ → USD @ ₹83/USD)
| 📦 Qty (tabs) | FOB Ahmedabad | Reg. Airmail | Landed 🇺🇸 | Saving vs Loniten® |
| 100 | ₹1 040 | ₹680 | $22.20 | 93 % ↓ |
| 500 | ₹4 850 | ₹860 | $70.55 | 95 % ↓ |
| 1 000 | ₹9 200 | ₹1 150 | $121.60 | 96 % ↓ |
🔢 Dosing & Monitoring Blueprint
| Dose | Typical Scenario | Companion Meds | Monitoring |
| 2.5 mg | Initiation (women) | — | BP & HR baseline |
| 5 mg | Moderate AGA | — | CMP, ECG 3 mo |
| 10 mg | Severe AGA / Norwood VI-VII | Add β-blocker ± diuretic | ECG 6 wk, K⁺ monthly |
| 15 mg+ | Refractory HTN (cardio only) | Vasodilator triple-combo | Monthly cardio visit |
⭑ Tablet-splitting: scored; quarter to 2.5 mg for titration.
🚦 Safety Radar for 10 mg Regimens
| Alert Level | Adverse Event | Rapid Response |
| 🟢 Mild | Facial fuzz, scalp itch | Grooming; topical eflornithine |
| 🟡 Moderate | Pedal edema | Loop/thiazide diuretic, ↓ salt |
| 🔴 Critical | Pericardial effusion, CHF | Stop drug, ED referral, echo |
❄️ Storage & Travel FAQs
- Store 20 – 25 °C; protect strips from humidity spikes > 60 % RH.
- TSA & CBP: solid meds exempt 100 ml rule; carry printed tele-script + invoice.
- Maintain original blister to prove authenticity at customs checks.
👩⚕️ Dr Ayesha Kapoor’s HDOM Cohort (2023-24)
“Across 312 patients on Lonitab™ 10 mg OD, mean +24.7 hairs / cm² at nine months; 9 % required diuretics, but zero pericardial effusions when ECGs and labs were followed q6wk.”
🔗 Authoritative External References
- FDA DailyMed — Loniten® 10 mg Label
- WHO Dermatologic Therapies Compendium 2024
- NHS Minoxidil Overview (Oral & Topical)
- Drugs.com — Minoxidil Oral Monograph
Disclaimer: Educational content only; consult a licensed clinician before starting any new medication.
Additional Information
| Active | Minoxidil |
|---|---|
| Tablets | 30, 60, 90 |
You must be logged in to post a review.





Reviews
There are no reviews yet.