- Your cart is empty
- Continue Shopping
✅ Active Ingredient Overview Table:
| 🔬 Active Ingredient (Generic Name): | Darifenacin Hydrobromide |
|---|---|
| 💊 Indication: | Overactive Bladder (OAB): Urgency, Frequency, Urge Incontinence |
| 🇺🇸 US Brand: | Enablex |
| 🏭 Manufacturer: | Cipla Limited (Founded: 1935) |
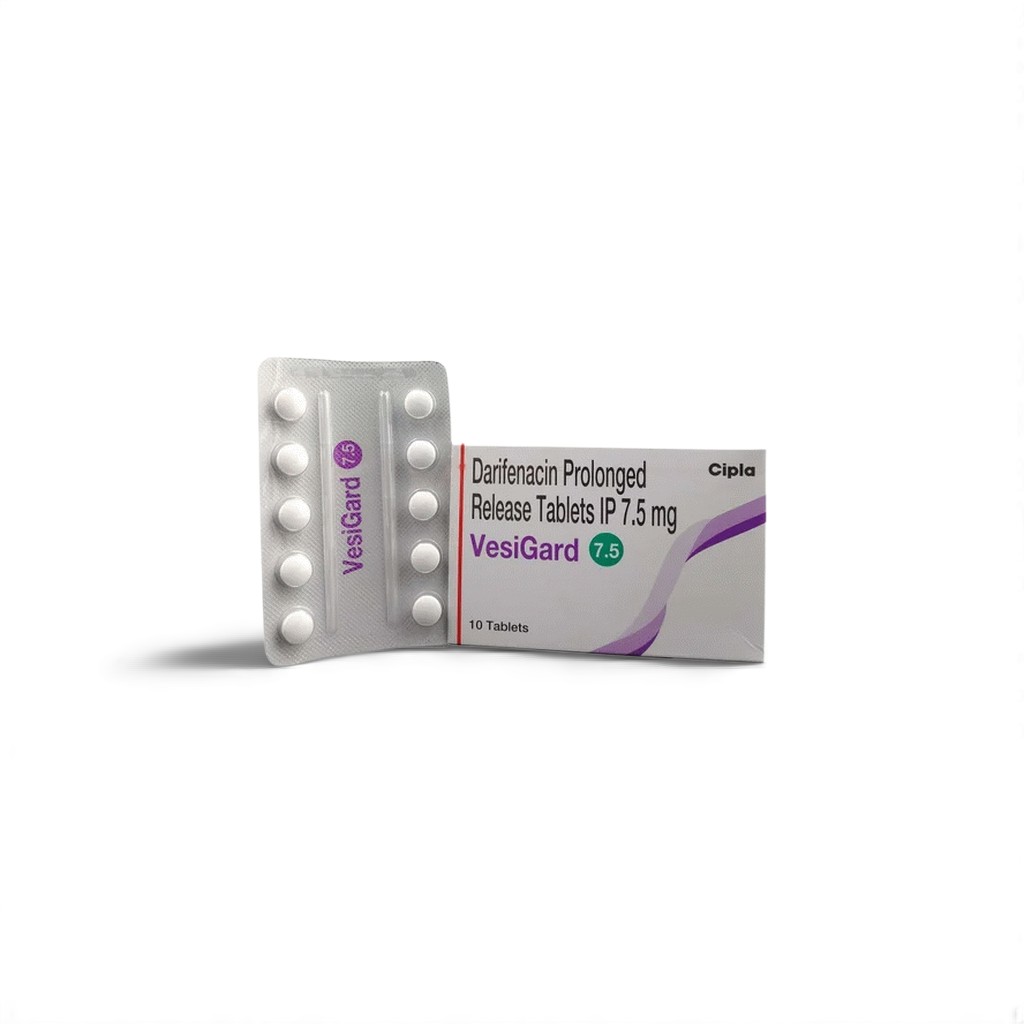
| 📦 Packaging: | 10 Tablets per Strip |
| 🧪 Strength: | 7.5 mg |
| 🚚 Delivery Time: | 6–15 days |
🌿 What is Darifenacin 7.5 mg (Vesigard)?
Vesigard 7.5 mg offers a targeted therapy for overactive bladder (OAB) by selectively antagonizing M3 muscarinic receptors in the detrusor muscle. Unlike nonselective antimuscarinics, Darifenacin’s high M3 receptor affinity leads to potent reduction of involuntary bladder contractions, resulting in fewer urgency and incontinence episodes.
Overactive bladder significantly impacts daily life—patients often experience frequent trips to the bathroom, sudden urges that disrupt activities, and social embarrassment. Early intervention with Vesigard can restore confidence, reduce nighttime awakenings, and improve overall quality of life.
Clinical studies demonstrate that a 7.5 mg daily dose of Darifenacin reduces daily incontinence episodes by up to 55% and decreases micturition frequency by an average of 2 episodes per day. Patients report improved sleep and reduced anxiety related to bladder control after four weeks of therapy.
For comprehensive prescribing and safety information, refer to the FDA label for Enablex, the Drugs.com monograph, the NHS Medicines A–Z entry, and the WHO Essential Medicines List.
🧬 Pharmacology & Mechanism of Action
| Attribute | Detail |
|---|---|
| Chemical Formula | C27H32N2O2•HBr |
| Molecular Weight | 516.47 g/mol (hydrobromide salt) |
| Bioavailability | ~15–19% |
| Metabolism | Hepatic (CYP2D6, CYP3A4) |
| Half-Life | ~13–19 hours |
Darifenacin’s M3 selectivity minimizes blockade of salivary and other muscarinic receptors, resulting in a favorable side effect profile.
💡 Indications & Dosing
| Condition | Dose & Administration |
|---|---|
| Overactive Bladder Symptoms | 7.5 mg once daily; swallow whole with water; may be taken with or without food |
A dose increase to 15 mg may be considered after 2–4 weeks in patients tolerating treatment but with insufficient symptom control. Dose reduction not typically required.
⚠️ Warnings & Side Effects
Common:
- Dry mouth
- Constipation
- Blurred vision
Serious:
- Urinary retention
- Gastrointestinal obstruction
- QT prolongation in high-risk patients
Contraindicated in urinary retention, gastric retention, uncontrolled narrow-angle glaucoma, and severe hepatic impairment. Monitor for anticholinergic adverse effects and cardiac conduction in predisposed patients.
🏥 Storage Instructions
Store Vesigard 7.5 mg at 20–25°C in original blister packaging. Protect from moisture and light. Keep out of reach of children.
🌐 Availability & Purchasing
- USA: Prescription-only (Enablex); branded and generic forms available.
- UK & EU: Prescription required; generic darifenacin accessible.
- Australia: Prescription and TGA approval required.
Importing from India may reduce costs; ensure regulatory compliance and valid prescription.
🔗 Related OAB Therapies
- Solifenacin 5 mg & 10 mg – Soliact by Cipla.
- Tolterodine ER 4 mg – Terol LA.
- Mirabegron 50 mg – β3-agonist.
🎥 Educational Video on Darifenacin
👩⚕️ Expert Insight: Dr. Kavita Desai, Urologist
“Darifenacin 7.5 mg strikes an excellent balance between efficacy and tolerability. It is a valuable first-line oral agent for moderate OAB symptoms.”
Additional Information
| Active | Darifenacin |
|---|---|
| Tablets | 30, 60, 90 |