- Your cart is empty
- Continue Shopping
✅ Active Ingredient Overview Table:
| 🔬 Active Ingredient (Generic Name): | Darifenacin Hydrobromide |
|---|---|
| 💊 Indication: | Overactive Bladder (OAB): Urgency, Frequency, Urge Incontinence |
| 🇺🇸 US Brand: | Enablex |
| 🏭 Manufacturer: | Cipla Limited (Founded: 1935) |
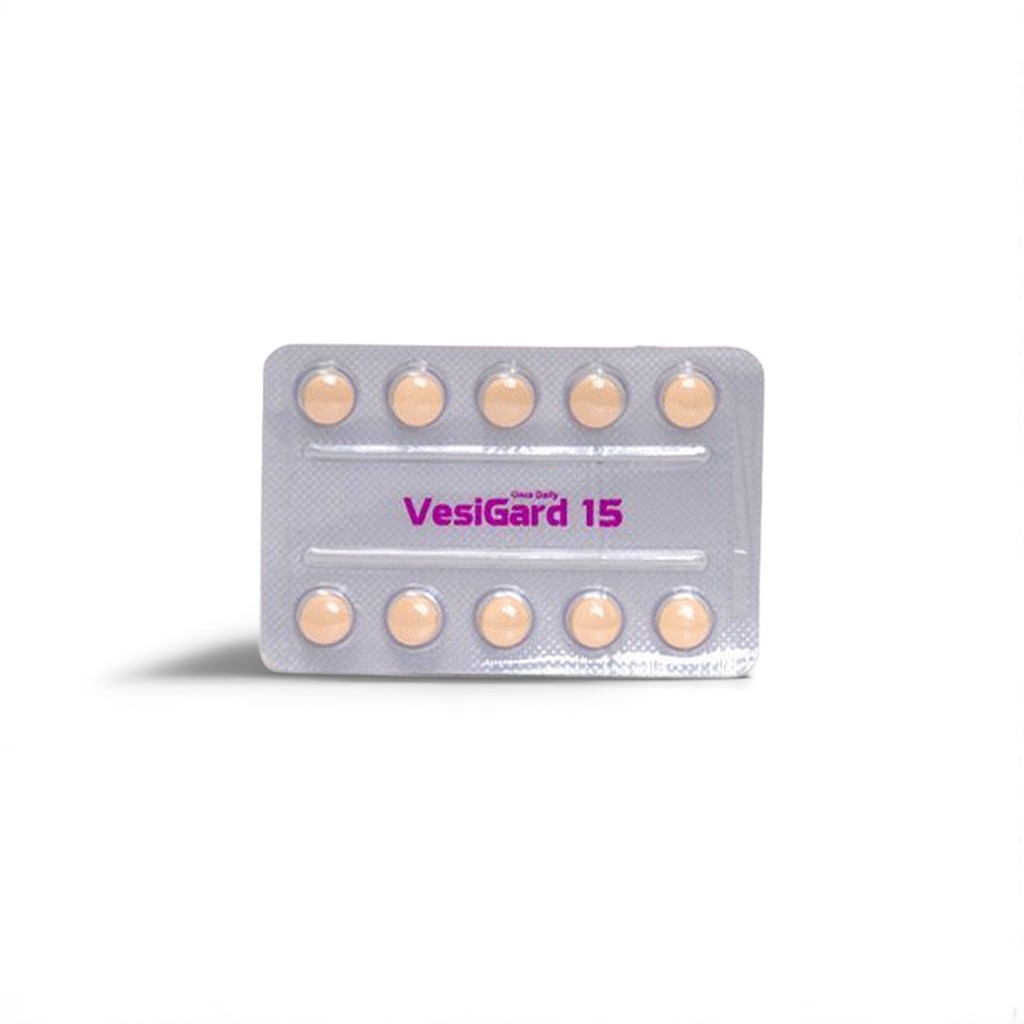
| 📦 Packaging: | 10 Tablets per Strip |
| 🧪 Strength: | 15 mg |
| 🚚 Delivery Time: | 6–15 days |
🌿 What is Darifenacin 15 mg (Vesigard)?
Vesigard 15 mg offers a higher potency option for patients with persistent OAB symptoms unresponsive to lower doses. Darifenacin selectively targets the M3 muscarinic receptor in bladder smooth muscle, reducing involuntary contractions, urgency, and leakage with sustained once-daily effects.
Overactive bladder can profoundly disrupt daily routines, sleep, and emotional well-being. For those whose symptoms continue despite 7.5 mg or 10 mg dosing, the 15 mg strength provides enhanced symptom reduction—clinical data show a further 15–20% decrease in incontinence episodes compared to the 7.5 mg dose, along with improved bladder capacity and patient-reported comfort.
Refer to the FDA label for Enablex, the Drugs.com monograph, the NHS entry, and the WHO Model List for complete prescribing details.
🧬 Pharmacology & Mechanism of Action
| Attribute | Detail |
|---|---|
| Chemical Formula | C27H32N2O2•HBr |
| Molecular Weight | 516.47 g/mol (hydrobromide salt) |
| Bioavailability | ~15–19% |
| Metabolism | Hepatic (CYP2D6, CYP3A4) |
| Half-Life | ~13–19 hours |
Darifenacin’s high M3 receptor affinity in the bladder reduces off-target cholinergic effects, even at elevated doses.
💡 Indications & Dosing
| Condition | Dose & Administration |
|---|---|
| Overactive Bladder Symptoms | 15 mg once daily with water; may be taken with or without food |
Initiate at 7.5 mg; increase to 15 mg after 2–4 weeks if symptoms persist and side effects are tolerated. No further dose escalation recommended.
⚠️ Warnings & Side Effects
Common:
- Dry mouth
- Constipation
- Blurred vision
Serious:
- Urinary retention
- Gastric retention
- QT prolongation (rare)
Contraindicated in patients with urinary or gastric retention, uncontrolled narrow-angle glaucoma, and severe hepatic impairment. Monitor for anticholinergic toxicity and adjust dose in elderly.
🏥 Storage Instructions
Store Vesigard 15 mg at 20–25°C in the original blister pack. Protect from moisture and light. Keep out of reach of children.
🌐 Availability & Purchasing
- USA: Prescription-only (Enablex 15 mg); branded and generics.
- UK & EU: Prescription required; generic darifenacin available.
- Australia: Prescription and TGA approval required.
Importing from India may lower costs; verify local regulations and maintain prescription.
🔗 Related Overactive Bladder Therapies
- Vesigard 7.5 mg – lower dose.
- Terol LA 4 mg – tolterodine ER.
- Mirabegron 50 mg – β3-adrenoceptor agonist.
🎥 Educational Video on Darifenacin
👩⚕️ Expert Insight: Dr. Kavita Desai, Urologist
“For patients with suboptimal response at lower strengths, Darifenacin 15 mg offers meaningful symptom relief. Continuous monitoring ensures safety and maximizes quality-of-life improvements.”
Additional Information
| Active | Darifenacin |
|---|---|
| Tablets | 30, 60, 90 |