- Your cart is empty
- Continue Shopping
✅ Active Ingredient Overview Table:
| 🔬 Active Ingredient (Generic Name): | Ceftriaxone Sodium |
|---|---|
| 💊 Indication: | Bacterial Infections (Sepsis, Meningitis, Pneumonia, Skin & Soft Tissue, Urinary) |
| 🇺🇸 US Brand Equivalent: | Rocephin |
| 🏭 Manufacturer: | Aristo Pharmaceuticals Pvt. Ltd (Founded: 1984) |
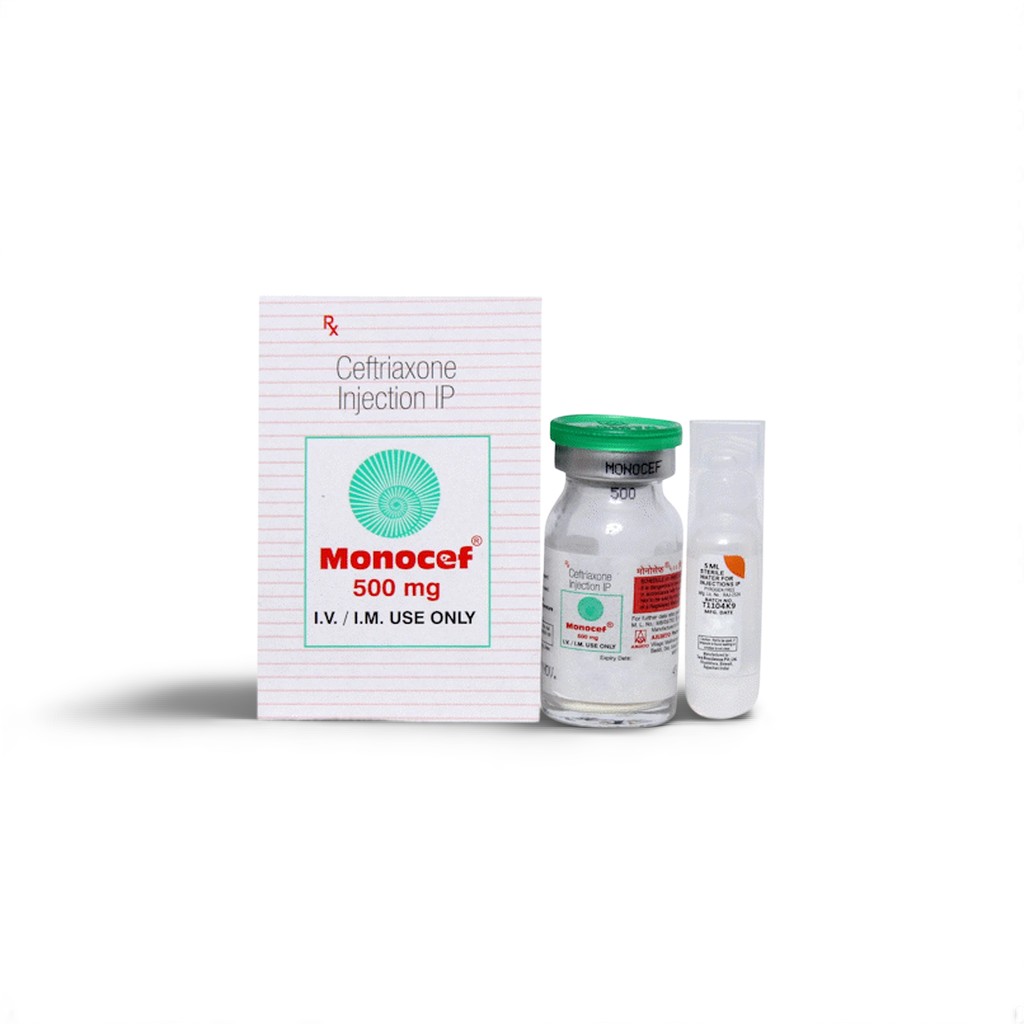
| 📦 Packaging: | 500 mg Ceftriaxone per 5 ml Vial |
| 🧪 Strength: | 500 mg (Reconstituted with Sterile Water for Injection) |
| 🚚 Delivery Time: | 6–15 days |
🌿 What is Monocef Injection 500 mg?
Monocef Injection 500 mg contains Ceftriaxone, a third-generation cephalosporin antibiotic renowned for its broad-spectrum activity against both Gram-positive and Gram-negative bacteria. As one of the most prescribed intravenous (IV) antibiotics worldwide, Ceftriaxone’s robust effectiveness stems from its ability to penetrate well into body tissues, including cerebrospinal fluid, making it a first-line agent for severe infections like bacterial meningitis, sepsis, and complicated pneumonia.
Moreover, Ceftriaxone exhibits high stability against most beta-lactamases, enzymes produced by resistant bacterial strains. Its long half-life (approximately 8 hours in adults) enables convenient once- or twice-daily dosing, which reduces nursing time and ensures consistent therapeutic levels. Monocef 500 mg is particularly valuable in hospital settings for treating severe infections, guiding empirical therapy in critically ill patients, and simplifying outpatient parenteral antibiotic therapy (OPAT).
When available for purchase in the US, UK, and Australia, clinicians and patients can leverage Monocef’s cost-effectiveness relative to branded equivalents. Ensuring a prescription and verifying authenticity through batch certificates are essential steps when sourcing Monocef 500 mg, whether domestically or via importation from India.
🧬 Pharmacology & Mechanism of Action
| Attribute | Detail |
|---|---|
| Chemical Formula | C18H18N8O7S3•Na (as sodium salt) |
| Molecular Weight | 641.47 g/mol (Ceftriaxone sodium) |
| Mechanism of Action | Binds to penicillin-binding proteins (PBPs) → inhibits peptidoglycan cross-linking → bactericidal |
| Bioavailability | 100% (IV/IM administration) |
| Half-Life | ~8 hours in adults; extended in neonates and in renal/hepatic impairment |
| Metabolism & Excretion | Excreted unchanged in urine (50–60%) and bile (40–50%); no dose adjustment needed for mild-to-moderate renal impairment; avoid calcium interactions in neonates |
💡 Indications & Dosage
| Condition | Dose & Administration |
|---|---|
| Community-Acquired Pneumonia (Adults) | 1–2 g IV once daily or 500 mg every 12 hours for 7–14 days based on severity and pathogen |
| Meningitis (Adults & Children ≥1 month) | 2 g IV every 12 hours (total 4 g/day) for 10–14 days; adjust for neonates |
| Sepsis (Adults) | 2 g IV once daily; may increase to 2 g every 12 hours in severe cases; combine with Gram-positive coverage (e.g., vancomycin) |
| Skin & Soft Tissue Infections (Adults) | 1–2 g IV once daily or 500 mg every 12 hours for 7–10 days |
| Uncomplicated Urinary Tract Infections (Adults) | 1 g IV once daily for 3 days |
| Gonorrhea (Adults) | 250 mg IM single dose plus single dose azithromycin 1 g orally (CDC dual therapy) |
| Meningitis (Neonates 0–28 days) | 50–75 mg/kg IV once daily (divided into two doses for neonates 0–14 days); see pediatric guidelines |
- Reconstitution: Add 9.6 ml Sterile Water for Injection to 500 mg vial → yields 500 mg/5 ml solution.
- IM Injection: Use a 21G needle; divide IM dose between two sites to reduce pain.
- IV Injection: Administer over 3–5 minutes; if dose ≥1 g, infuse over 30 minutes to minimize infusion-related reactions.
- Duration: Determined by infection type and clinical response; follow institutional or guideline recommendations.
⚠️ Warnings & Side Effects
Common Side Effects:
- Injection site pain or induration
- Gastrointestinal: Diarrhea, nausea, vomiting
- Rash, urticaria
Less Common:
- Transient elevated liver enzymes
- Eosinophilia
Serious (Rare):
- Clostridioides difficile–associated diarrhea (CDAD)
- Hemolytic anemia, thrombocytopenia
- Severe hypersensitivity reactions (anaphylaxis, serum sickness–like reactions)
Precautions:
- Hypersensitivity: Contraindicated in patients with known allergy to cephalosporins.
- Neonates: Avoid IV calcium-containing solutions with Ceftriaxone due to precipitation risk in neonates (≤28 days).
- Renal/Hepatic Impairment: No adjustment for mild-to-moderate dysfunction; monitor in severe cases.
- Clostridioides difficile: Use caution; monitor for diarrhea.
- Calcium Interaction: Do not co-administer or mix with calcium-containing IV solutions in neonates; separate calcium infusion by ≥48 hours after Ceftriaxone dose.
🩺 Storage Instructions
Store Monocef 500 mg vials at 20–25°C (68–77°F). Protect from light. Once reconstituted, use within 6 hours at room temperature or within 24 hours if refrigerated (2–8°C). Discard any unused portion accordingly. Keep out of reach of children.
🌐 Availability & Purchasing
Monocef 500 mg is available by prescription in various markets. When considering purchase, the following keywords can assist in locating reliable suppliers.
- Buy in US: Search for “Monocef 500 mg injection” or “Ceftriaxone 500 mg vial” from licensed pharmacies. Prescription required.
- Buy in UK: Look for “Monocef 500 mg” or “Ceftriaxone 500 mg” via NHS or private clinics. Prescription mandatory for antibiotic therapy.
- Buy in Australia: Use terms “Monocef 500 mg” or “Ceftriaxone 500 mg” through TGA-approved distributors; import permitted with valid prescription.
Importing from India:
Purchasing Monocef 500 mg Injection directly from India can significantly reduce costs. Ensure compliance with import regulations (FDA personal import scheme, UK MHRA guidelines, TGA permits), maintain a valid prescription, and verify product authenticity with batch certificates to avoid counterfeit antibiotics.
🔗 Authoritative Resources
🔗 Antibiotic Injections
- Ceftriaxone 250 mg Injection – Generic Ceftriaxone
🎥 Educational Video on Ceftriaxone Use & Safety
👨⚕️ Expert Insight: Dr. Rajiv Mehta, Infectious Disease Specialist
“Ceftriaxone’s long half-life and extensive tissue penetration make Monocef 500 mg Injection a cornerstone for empiric and definitive therapy in severe gram-negative and gram-positive infections. Clinicians should balance dosage with patient renal and hepatic function to optimize outcomes.”
Additional Information
| Active | Ceftriaxone |
|---|---|
| Injections/s | 1, 3, 6 |