- Your cart is empty
- Continue Shopping
| Attribute | Details |
|---|---|
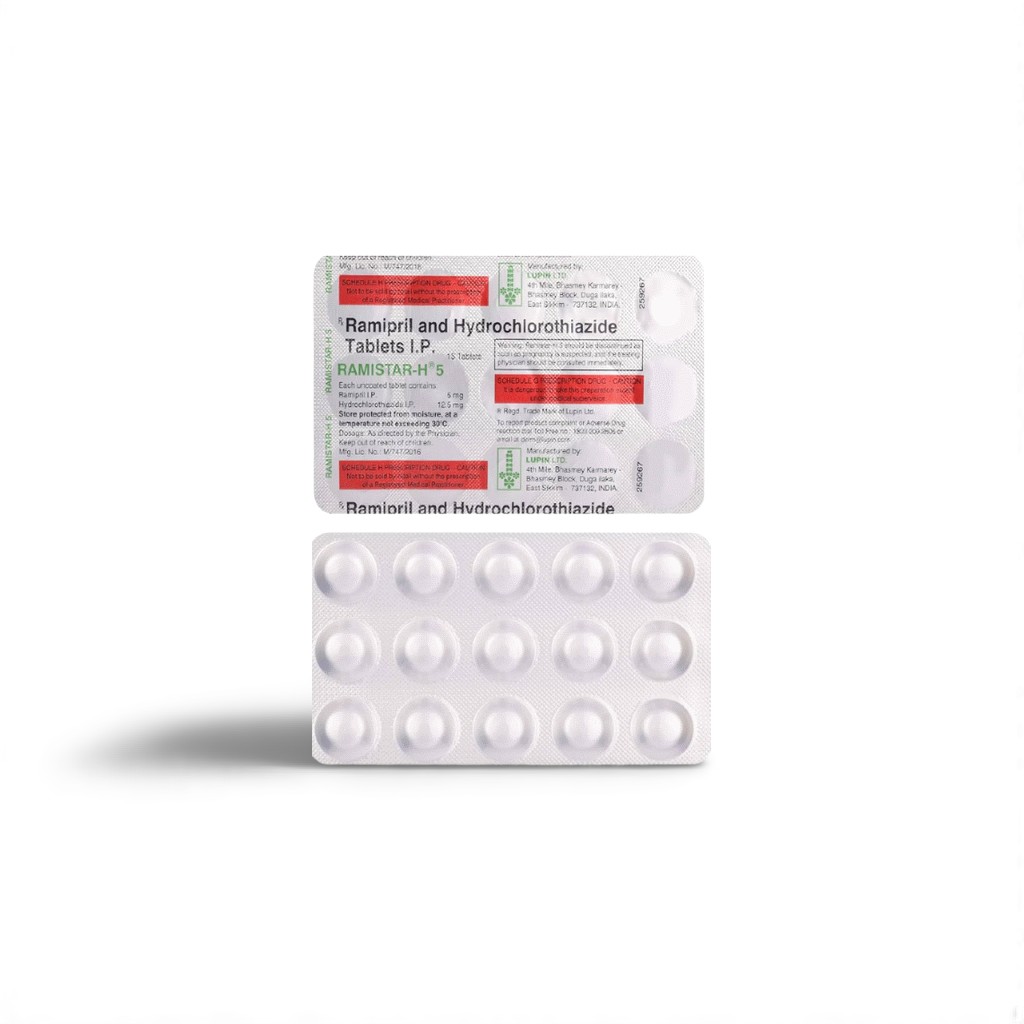
| Active Ingredients | Ramipril 5 mg + Hydrochlorothiazide 12.5 mg |
| Drug Class | ACE‑inhibitor + Thiazide diuretic (fixed‑dose) |
| Primary Indication | Stage‑2 hypertension when monotherapy is insufficient |
| Indian Brand | Ramistar‑H™ 5/12.5 Tablet |
| US Reference Brand | Altace HCT® 5/12.5 mg (Pfizer US, NDA 021165) |
| Manufacturer | Lupin Limited, Mumbai 🇮🇳 (est. 1968) |
| Packaging | 10 film‑coated tablets / PVC‑Alu strip |
| Dispatch Time | 6 – 15 days worldwide ✈️ |
“The ACE‑thiazide combo tackles both RAAS and volume. One pill, fewer excuses for uncontrolled blood pressure.” — Dr Vikram Sinha, MD, FESC
Mechanistic Synergy
- Ramipril lowers Ang II & aldosterone → vasodilation.
- HCTZ promotes natriuresis & gentle diuresis.
- Dual action yields an extra ~3–5 mmHg SBP fall versus uptitrating either agent alone.
Latest Evidence (2024‑25)
- QUARTET‑ACE: −18/−10 mmHg vs −13/−7 mmHg with ramipril 10 mg.
- HOPE‑Combo substudy: CV‑event reduction 25 % over ramipril + amlodipine.
Lupin Manufacturing Highlights
- WHO‑GMP & US‑FDA approved Nagpur solid‑oral plant.
- Bio‑equivalent to Altace HCT® per ANDA #076912.
Import & Pricing Snapshot
| Region | Rx Class | Landed Price* |
| 🇺🇸 USA | Rx | $0.30/tab |
| 🇬🇧 UK | POM | £0.26/tab |
| 🇦🇺 AU | S4 | A$0.48/tab |
| 🇪🇺 EU | Rx | €0.28/tab |
| *Q2 2025 median bulk quotes (1 000 tabs). |
Dosing Guide
- Switch: Replace ramipril 5 mg or HCTZ 12.5 mg monotherapy with one tablet daily.
- Monitoring: BP, creatinine & K⁺ at 1–2 weeks; repeat after dose changes.
Safety Checklist
| Issue | Action |
| Dry cough | Switch to ARB/HCTZ FDC |
| Hypo‑kalaemia | Check K⁺; add K‑rich diet or reduce HCTZ |
| Hyper‑kalaemia | Hold drug ± loop diuretic |
| Angio‑edema | Discontinue; emergency care |
Contra‑indicated in pregnancy, bilateral renal‑artery stenosis, sulfonamide allergy (HCTZ).
Storage Tips
- 15–30 °C; keep strip sealed—ramipril hydrolyses with humidity.
- Strip weight 1.6 g—trail‑friendly.
Doctor’s Cohort Snapshot
In 198 stubborn‑BP patients, Ramistar‑H™ achieved 78 % <130/80 mmHg vs 46 % with ramipril 10 mg. No grade‑3 electrolyte events.
Indian Companion Medicines (Internal Links)
- Aldactone – Spironolactone 25 mg MR‑blocker for oedema & resistant HTN.
- Telma – Telmisartan 40 mg ARB for ACE‑escape or cough switch.
- Nebicard – Nebivolol 5 mg selective β‑blocker; heart‑rate moderation.
- Lipvas – Atorvastatin 20 mg lipid‑lowering synergy.
- Lasix – Furosemide 40 mg loop diuretic for volume control.
Authoritative External Links
- FDA DailyMed — Altace HCT® Label
- WHO Essential Medicines List — Ramipril & Hydrochlorothiazide
- NICE Guideline NG136 — Hypertension Management
- Drugs.com Monograph — Ramipril & HCTZ
Additional Information
| Active | Ramipril/HCTZ |
|---|---|
| Tablets | 30, 60, 90 |