- Your cart is empty
- Continue Shopping
| 🌐 Specification | 🧾 Details |
|---|---|
| 💊 Active Ingredient | Methylprednisolone Sodium Succinate |
| 🧪 Strength | 125 mg |
| 🏭 Manufacturer | Pfizer India Ltd |
| 💉 Form | Injection (Lyophilized Powder) |
| 📦 Packaging | 2 ml in 1 vial |
| ⏱️ Delivery Time | 6 to 15 days |
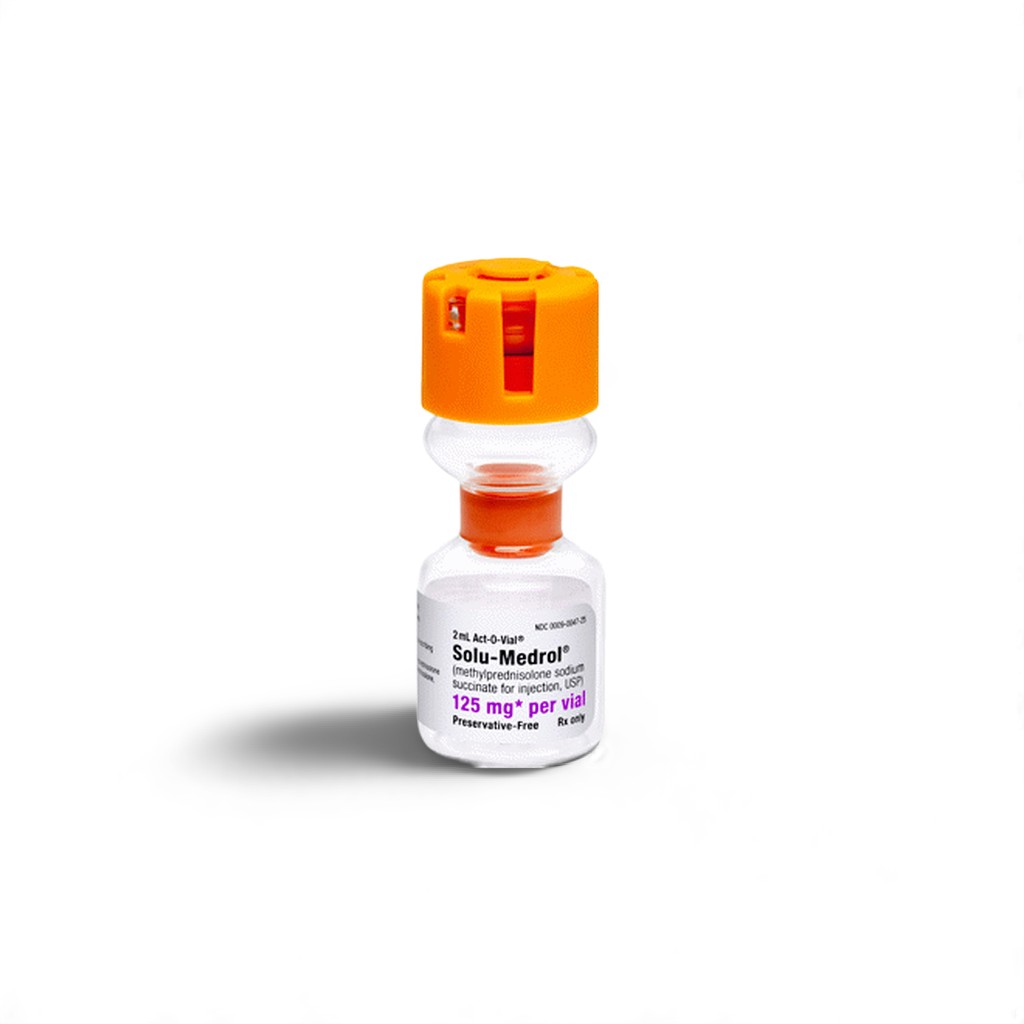
🧬 What is Solu-Medrol 125mg Used For?
Solu-Medrol 125 mg is an injectable corticosteroid used for rapid control of severe inflammation in acute medical situations. It contains methylprednisolone sodium succinate, a fast-acting glucocorticoid administered intravenously or intramuscularly in emergencies.
Manufactured by Pfizer India Ltd., this preparation is widely used globally in emergency rooms, ICUs, and ambulatory care settings for quick symptom control in life-threatening conditions. It is ideal for patients experiencing sudden exacerbations of asthma, anaphylaxis, or autoimmune flares.
🎥 Educational Video: Administering Solu-Medrol Safely
📺 “Solu-Medrol 125mg Injection – Emergency Corticosteroid Therapy Explained”
🎬 Video Placeholder — Covers preparation, dosage, and monitoring during administration.
Always seek a doctor’s supervision when using injectable corticosteroids.
🩺 Indications for Use
- Acute asthma or COPD exacerbations
- Anaphylaxis (as part of multi-drug protocol)
- Multiple sclerosis relapses
- Acute gout or arthritis
- Systemic lupus erythematosus
- Organ transplant rejection episodes
- Acute spinal cord injury (in high doses)
💉 Dosage & Administration
| 🏥 Clinical Condition | 💉 Typical Dose |
|---|---|
| Acute asthma or allergic crisis | 125 mg IV/IM once |
| Autoimmune flare-up | 125–500 mg IV per day |
| Spinal cord trauma (initial) | Up to 30 mg/kg IV (special protocol) |
The dose may vary based on medical urgency. IV administration preferred for immediate response.
⚠️ Side Effects and Precautions
| 💥 Common Effects | ❗ Serious Risks |
|---|---|
| Insomnia, fluid retention | Infection risk, GI bleeding, adrenal crisis |
| Blood sugar elevation | Cushing’s syndrome, psychiatric changes |
| Facial puffiness | Cardiovascular collapse (rare) |
🧃 Storage Instructions
- Store between 20–25°C
- Do not freeze the dry powder or solvent
- Use immediately after reconstitution
- Discard unused portions
🌍 Global Availability & Import Guide
| 🌎 Region | 📦 Import Eligibility | 📋 Prescription Rule |
|---|---|---|
| 🇺🇸 USA | Allowed via FDA personal use | Prescription mandatory |
| 🇬🇧 UK | Importable via MHRA guidelines | Rx required |
| 🇦🇺 Australia | Permitted by TGA rules | Prescription or medical note |
🔗 Related Corticosteroid Injectables from India
| 💊 Product Name | 🏭 Manufacturer | 📖 Description |
|---|---|---|
| Depo-Medrol Injection – Pfizer | Pfizer India | Long-acting methylprednisolone acetate for intramuscular use. |
| Wysolone Oral – Pfizer | Pfizer India | Oral prednisolone used for long-term inflammation control. |
| Omnacortil Injection – Macleods | Macleods Pharmaceuticals | Injectable corticosteroid alternative for systemic treatment. |
| Predmet Injection – Sun Pharma | Sun Pharmaceutical | Methylprednisolone acetate injection for allergy and arthritis. |
| Defcort Oral – Glenmark | Glenmark Pharmaceuticals | Oral deflazacort alternative, mild immunosuppressive steroid. |
📚 References & External Resources
- FDA Drug Database – Methylprednisolone
- Drugs.com – Solu-Medrol Information
- WHO – Corticosteroids for Emergency Use
- NHS – Corticosteroid Injections
🩺 Doctor’s Commentary – Dr. Aakash Deshmukh, MD Emergency Medicine
“Solu-Medrol 125mg is a critical intervention in any emergency toolkit. When seconds matter, its rapid absorption and broad anti-inflammatory activity can stabilize patients experiencing respiratory or autoimmune collapse.”
⚠️ Disclaimer
This content is for educational purposes only. Solu-Medrol 125mg should only be used under medical supervision. Follow country-specific regulations for importing corticosteroids.
Additional Information
| Active | Methylprednisolone |
|---|---|
| Injection/s | 1, 3, 6 |