- Your cart is empty
- Continue Shopping
| 🏷️ Parameter | 🔍 Details |
|---|---|
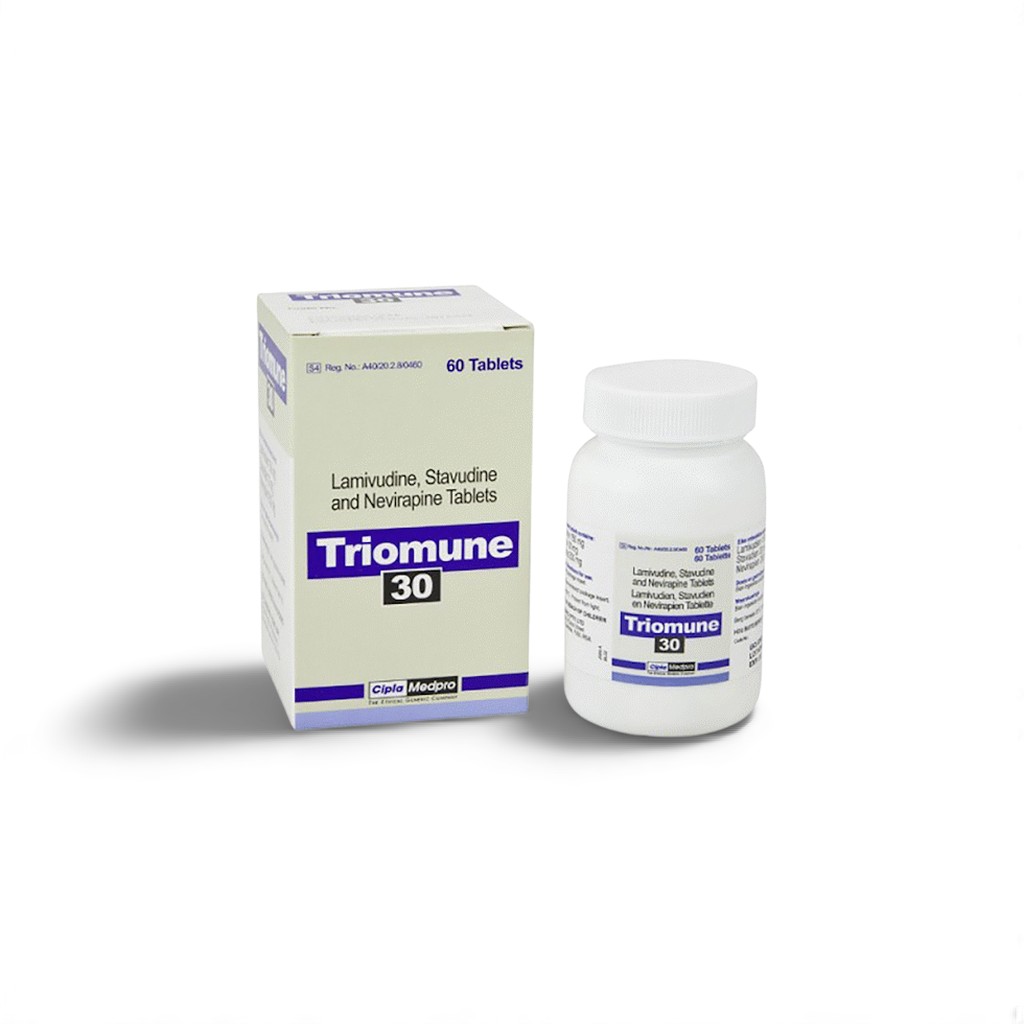
| 🧬 Active Ingredients | Stavudine 30mg + Lamivudine 150mg + Nevirapine 200mg |
| 💊 Indication | HIV-1 Infection (First-line ART regimen) |
| 🇺🇸 US Brand | No direct equivalent; components available separately |
| 🏭 Manufacturer | Cipla Ltd. |
| 📦 Packaging | Bottle 30 pills |
| 💪 Strength | 30mg/150mg/200mg per tablet |
| 🚚 Delivery Time | 7–14 days (Worldwide) |
🧬 Introduction to Triomune
Triomune by Cipla is a fixed-dose combination of three antiretrovirals:
-
⚡ Stavudine 30mg (NRTI: inhibits viral DNA synthesis)
-
🔒 Lamivudine 150mg (NRTI: chain terminator)
-
🔥 Nevirapine 200mg (NNRTI: blocks reverse transcriptase)
Approved as first-line therapy for HIV-1 in resource-limited settings, it offers:
-
✅ Simplified dosing (1 tablet BID after 14-day Nevirapine lead-in)
-
✅ Cost-effective viral suppression
-
❗ Black-box warnings for mitochondrial toxicity (Stavudine) and hepatotoxicity (Nevirapine)
Cipla’s formulation revolutionized global HIV access but is now phased out in developed countries due to toxicity concerns.
💊 Dosage & Indications
| 💉 Indication | 🧪 Dosage | ⏱️ Frequency | 💡 Notes |
|---|---|---|---|
| Adults >60kg | 1 tablet BID | Twice daily | *After 14-day Nevirapine lead-in (200mg/day)* |
| Adults <60kg | Not recommended | – | Use separate formulations |
| Children | Contraindicated | – | Use weight-adjusted doses |
| Renal Impairment | Avoid if CrCl <50 mL/min | Contraindicated | Lamivudine accumulation |
⚠️ Side Effects, Storage & Warnings
-
❗ Common Side Effects:
-
Peripheral neuropathy (Stavudine)
-
Rash (Nevirapine)
-
Nausea (Lamivudine)
-
-
🚫 Serious Effects:
-
Lactic acidosis (Stavudine)
-
Fatal hepatotoxicity (Nevirapine)
-
Pancreatitis (Stavudine)
-
-
❄️ Storage: 15–30°C; protect from moisture (keep in blister until use).
-
⚠️ Warnings:
-
Contraindicated in moderate-severe liver disease
-
Avoid in women with CD4>250/mm³ or men with CD4>400/mm³
-
Monitor for lipoatrophy (face/limb fat loss)
-
🌍 Global Availability & Shipping
| 🌎 Region | 📦 Shipping | 💵 Price (60 tablets) | 📜 Prescription |
|---|---|---|---|
| 🇺🇸 USA | ✅ Restricted | $55–$90 | Required |
| 🇬🇧 UK | ✅ Yes | £45–£75 | Required |
| 🇦🇺 Australia | ❌ No | – | Not available |
🔗 Related Indian Alternatives (Different in Same Category)
| 💊 Product Name | 🏭 Manufacturer | 🔗 Internal Link |
|---|---|---|
| Viraday (Efavirenz/Emtricitabine/Tenofovir) | Cipla | viraday |
| Doluteg-L (Dolutegravir/Lamivudine) | Emcure | doluteg-l |
| Tenvir-EM (Tenofovir/Emtricitabine) | Cipla | tenvir-em |
| Abamune-L (Abacavir/Lamivudine) | Cipla | abamune-l |
| Lopimune (Lopinavir/Ritonavir) | Cipla | lopimune |
| Covihope-D (Dolutegravir/Ritonavir) | Hetero | covihope-d |
📚 External Medical References
🩺 Expert Commentary
Dr. Rajiv Malhotra, HIV Researcher:
*”While Triomune expanded ART access globally, its Stavudine component causes irreversible lipoatrophy in 20% of patients. Modern regimens like [viraday] (Tenofovir-based) are safer first-line options. Reserve Triomune for salvage therapy when alternatives are unavailable.”*
📌 Disclaimer
This information is not medical advice. Use under physician supervision. Cipla’s Triomune carries FDA black-box warnings for hepatotoxicity and mitochondrial toxicity.
💡 Clinical Protocol:
-
Stavudine Toxicity Monitoring:
-
Baseline & quarterly: Lactate levels, liver function
-
Monthly: Peripheral sensation tests
-
-
Nevirapine Lead-in:
-
200mg/day × 14 days → if no rash, start Triomune BID
-
-
Cipla Tablet ID: “CIPLA” logo + “T30” on tablets
-
Discontinuation Criteria:
-
Lactate >5 mmol/L + symptoms
-
ALT >10× ULN
-
Severe lipoatrophy
-
-
Alternative Regimen: Switch to [tenvir-em] if weight loss >10% in limbs
Additional Information
| Active | Stavudine/Lamivudine/Nevirapine |
|---|---|
| Tablets | 30, 60, 90 |