- Your cart is empty
- Continue Shopping
| 🏷️ Parameter | 🔍 Details |
|---|---|
| 🧬 Active Ingredient | Abacavir Sulfate |
| 💊 Indication | HIV-1 Infection (part of combination ART) |
| 🇺🇸 US Brand | Ziagen® |
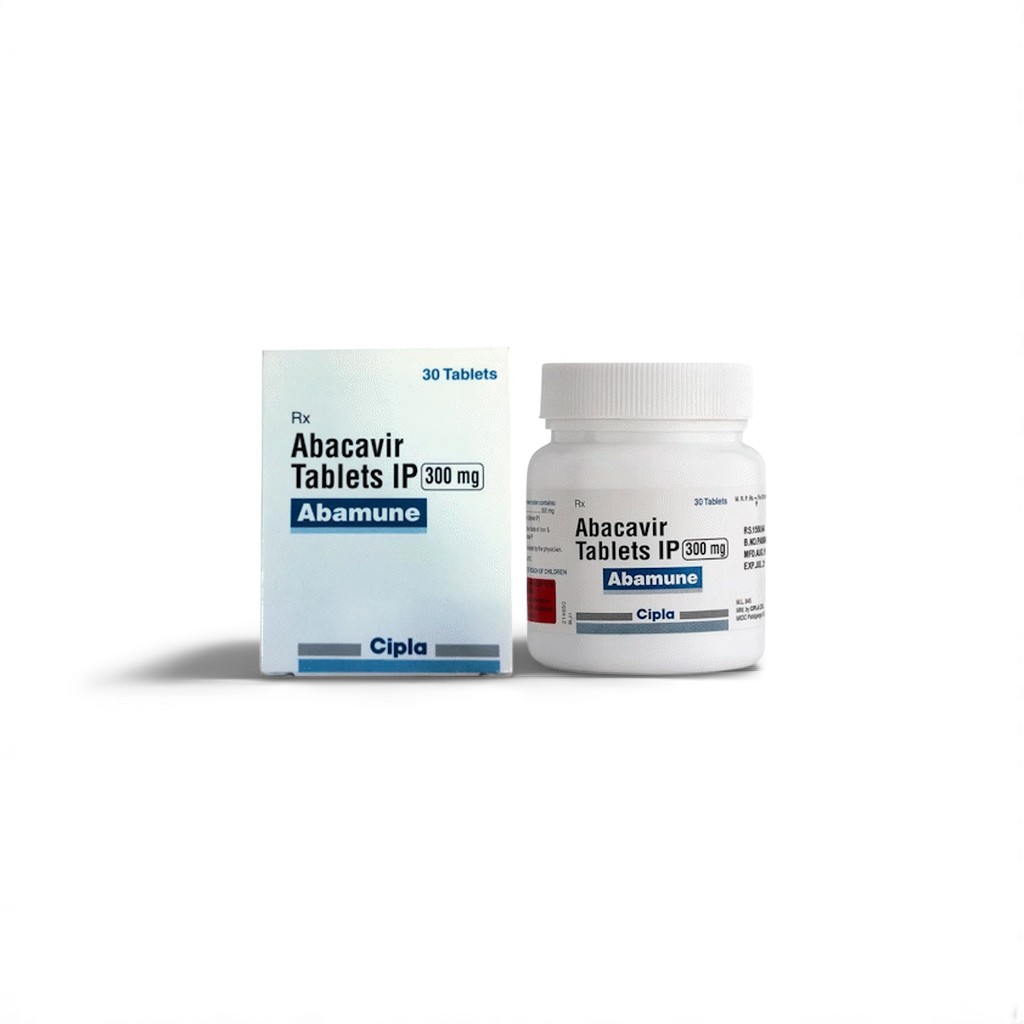
| 🏭 Manufacturer | Cipla Ltd. |
| 📦 Packaging | 30 tablets in 1 HDPE bottle |
| 💪 Strength | 300 mg |
| 🚚 Delivery Time | 7–14 days (Worldwide) |
⚙️ Introduction to Abamune
Abamune by Cipla contains Abacavir, a nucleoside reverse transcriptase inhibitor (NRTI) for HIV-1 treatment. Critical considerations:
-
🧬 Mandatory HLA-B*5701 screening before initiation (fatal hypersensitivity risk in positive patients)
-
❤️ Increased myocardial infarction risk in patients with cardiac risk factors
-
✅ Preferred for patients with renal impairment (no dose adjustment needed)
-
⚠️ Black-box warning for hypersensitivity reactions (fever, rash, multi-organ involvement)
Cipla’s 30-tablet bottle provides a month’s supply for combination regimens like Abacavir+Lamivudine+Dolutegravir.
💊 Dosage & Indications
| 💉 Indication | 🧪 Dosage | ⏱️ Frequency | 💡 Notes |
|---|---|---|---|
| Adults | 600 mg/day | Once daily or 300 mg BID | HLA-B5701 negative only* |
| Children ≥25kg | 300 mg BID | Twice daily | Oral solution for <25kg |
| Renal Impairment | No adjustment needed | – | Preferred over TDF |
| Hepatic Impairment | Avoid in severe cirrhosis | Contraindicated | ↓ Metabolism |
⚠️ Side Effects, Storage & Warnings
-
❗ Common Side Effects:
-
Nausea (19%)
-
Headache (13%)
-
Fatigue (9%)
-
-
🚫 Life-Threatening Effects:
-
Hypersensitivity reaction (fever, rash, respiratory failure)
-
Myocardial infarction (in high-risk patients)
-
Lactic acidosis
-
-
❄️ Storage: 15–30°C; keep bottle tightly closed (desiccant included).
-
⚠️ Critical Warnings:
-
Contraindicated without confirmed HLA-B*5701 negative status
-
Discontinue permanently if hypersensitivity suspected
-
Monitor cardiovascular risk factors biannually
-
🌍 Global Availability & Shipping
| 🌎 Region | 📦 Shipping | 💵 Price (30-tablet bottle) | 📜 Prescription |
|---|---|---|---|
| 🇺🇸 USA | ✅ Yes | $45–$80 | Required |
| 🇬🇧 UK | ✅ Yes | £35–£65 | Required |
| 🇦🇺 Australia | ✅ Yes | AUD 60–100 | Required |
🔗 Related Indian Alternatives (Different in Same Category)
| 💊 Product Name | 🏭 Manufacturer | 🔗 Internal Link |
|---|---|---|
| Tenvir (Tenofovir) | Cipla | tenvir |
| Lamivir (Lamivudine) | Cipla | lamivir |
| Viraday (Efavirenz/FTC/TDF) | Cipla | viraday |
| Taffic (Bictegravir/TAF/FTC) | Hetero | taffic |
| Dolutranz (Dolutegravir/Tenofovir) | Mylan | dolutranz |
| Abamune-L (Abacavir/Lamivudine) | Cipla | abamune-l |
📚 External Medical References
🩺 Expert Commentary
Dr. Priya Desai, HIV Pharmacologist:
“Abamune’s renal safety makes it ideal for older patients but demands strict HLA-B5701 screening. For those with cardiovascular risks, [tenvir] (Tenofovir) is safer. Cipla’s bottle includes a hypersensitivity warning card – ensure patients carry it at all times.”*
📌 Disclaimer
This information is not medical advice. Use under physician supervision. Cipla’s Abamune carries black-box warnings for fatal hypersensitivity reactions.
💡 Clinical Protocol:
-
Hypersensitivity Reaction (HSR) Management:
Symptom Onset Action Within 6 weeks Permanent discontinuation Fever + Rash ER visit immediately Respiratory symptoms Epinephrine + corticosteroids -
Cardiovascular Monitoring:
Parameter Frequency Action Threshold Blood Pressure Quarterly >140/90 mmHg Lipid Profile Biannual LDL >160 mg/dL ECG Baseline Ischemic changes -
Bottle Specifications:
-
Amber HDPE bottle with induction seal
-
Tablet ID: “ABA300” engraving
-
Hypersensitivity warning card included
-
-
Drug Interactions:
Concomitant Drug Risk Action Ethanol ↑ Abacavir levels 40% Avoid excessive alcohol Methadone No interaction Safe for MAT patients Ribavirin ↑ Lactic acidosis risk Avoid concurrent use -
HLA-B*5701 Screening Protocol:
-
Test before first prescription
-
Document negative result in medical record
-
Re-testing unnecessary if negative
-
Additional Information
| Active | Abacavir |
|---|---|
| Tablets | 30, 60, 90 |