- Your cart is empty
- Continue Shopping
| 🏷️ Parameter | 🔍 Details |
|---|---|
| 🧬 Active Ingredients | Stavudine 40mg + Lamivudine 150mg + Nevirapine 200mg |
| 💊 Indication | HIV-1 Infection (First-line ART for patients >60kg) |
| 🇺🇸 US Brand | No direct equivalent; components available separately |
| 🏭 Manufacturer | Cipla Ltd. |
| 📦 Packaging | 30 tablets in 1 HDPE bottle |
| 💪 Strength | 40mg/150mg/200mg per tablet |
| 🚚 Delivery Time | 7–14 days (Worldwide) |
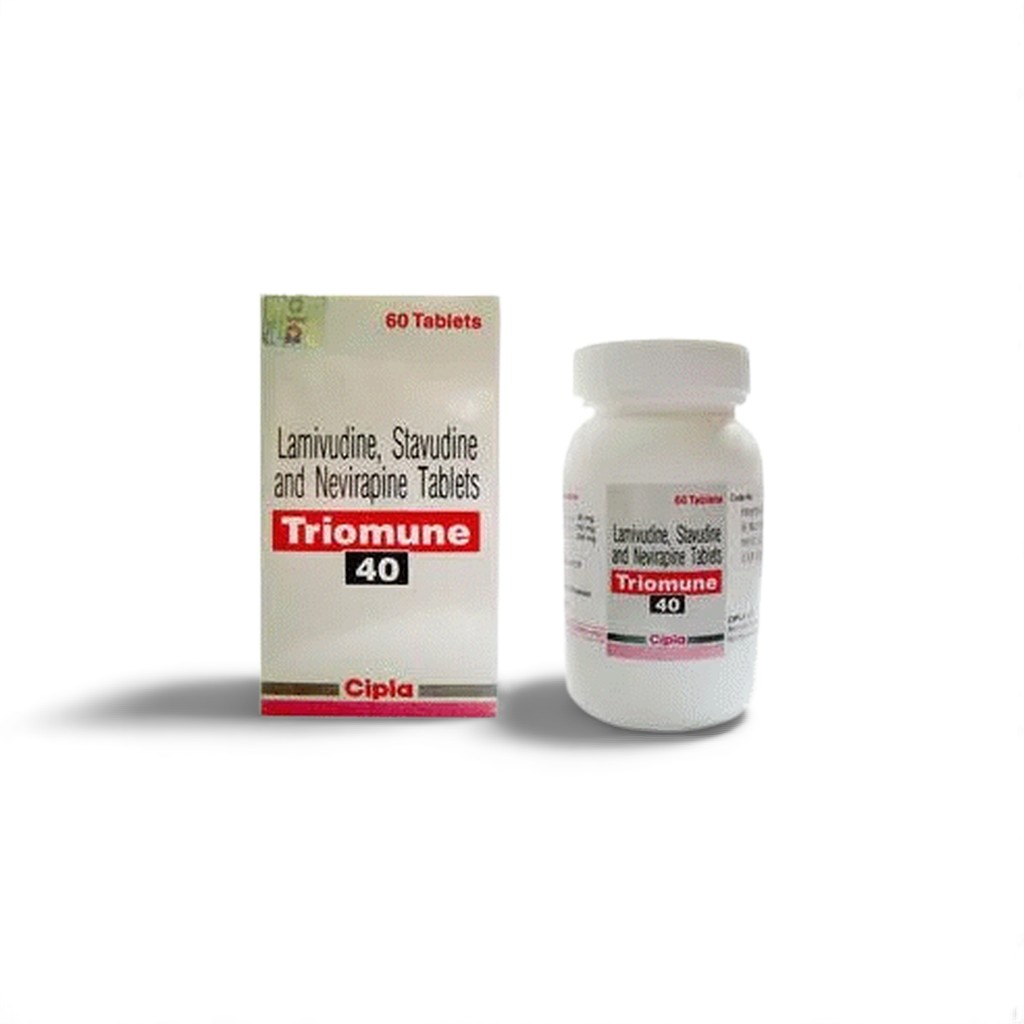
🧬 Introduction to Triomune 40mg/150mg/200mg
Triomune by Cipla is a high-dose fixed-dose combination for HIV-1 treatment, containing:
-
⚡ Stavudine 40mg (NRTI: higher dose for patients >60kg)
-
🔒 Lamivudine 150mg (NRTI: prevents viral replication)
-
🔥 Nevirapine 200mg (NNRTI: targets reverse transcriptase)
Designed for adult patients >60kg, this 30-tablet bottle provides a 15-day maintenance supply after the lead-in phase. Key considerations:
-
⚠️ 30% higher risk of peripheral neuropathy vs. 30mg formulation
-
✅ Cost-effective for resource-limited settings
-
❗ Phased out in high-income countries due to toxicity concerns
💊 Dosage & Indications
| 💉 Indication | 🧪 Dosage | ⏱️ Frequency | 💡 Notes |
|---|---|---|---|
| Adults >60kg (Maintenance) | 1 tablet BID | Twice daily | *After 14-day Nevirapine lead-in (200mg/day)* |
| Adults <60kg | Contraindicated | – | Use Triomune 30mg formulation |
| Lead-in Phase | Nevirapine 200mg/day alone | Once daily | 14 days (monitor rash) |
| Renal Impairment (CrCl <50) | Avoid | Contraindicated | Lamivudine accumulation risk |
⚠️ Side Effects, Storage & Warnings
-
❗ Common Side Effects:
-
Numbness/tingling (42% at 40mg dose)
-
Hyperlactatemia (elevated lactate)
-
Rash (25% during lead-in)
-
-
🚫 Serious Effects:
-
Lactic acidosis (fatal in 0.8% of cases)
-
Severe hepatotoxicity
-
Pancreatic inflammation
-
-
❄️ Storage: 15–30°C; keep bottle tightly closed (desiccant included).
-
⚠️ Critical Warnings:
-
Contraindicated if baseline lactate >2 mmol/L
-
Monthly monitoring: Liver enzymes + lactate levels
-
Permanent discontinuation for lipoatrophy (face/limb fat loss)
-
🌍 Global Availability & Shipping
| 🌎 Region | 📦 Shipping | 💵 Price (30-tablet bottle) | 📜 Prescription |
|---|---|---|---|
| 🇺🇸 USA | ✅ Restricted | $65–$100 | Required |
| 🇬🇧 UK | ✅ Yes | £50–£80 | Required |
| 🇦🇺 Australia | ❌ No | – | Not available |
🔗 Related Indian Alternatives (Different in Same Category)
| 💊 Product Name | 🏭 Manufacturer | 🔗 Internal Link |
|---|---|---|
| Viraday (Efavirenz/Emtricitabine/Tenofovir) | Cipla | viraday |
| Dolutranz (Dolutegravir/Tenofovir) | Mylan | dolutranz |
| Tenof-EM (Tenofovir/Emtricitabine) | Hetero | tenof-em |
| Abacavir-L (Abacavir/Lamivudine) | Cipla | abacavir-l |
| Lopimune (Lopinavir/Ritonavir) | Cipla | lopimune |
| Covihope-D (Dolutegravir/Ritonavir) | Hetero | covihope-d |
📚 External Medical References
🩺 Expert Commentary
Dr. Sanjay Verma, HIV Pharmacologist:
*”The 40mg Stavudine in Triomune significantly increases neuropathy risk compared to 30mg. For patients >60kg, I recommend transitioning to [tenof-em] (Tenofovir-based regimen) within 6 months. Always conduct monthly lactate checks – Cipla’s bottle includes desiccant to maintain stability in tropical climates.”*
📌 Disclaimer
*This information is not medical advice. Use under physician supervision. Cipla’s Triomune 40mg carries black-box warnings for fatal lactic acidosis and hepatotoxicity.*
💡 Clinical Protocol:
-
Toxicity Monitoring Schedule:
Test Frequency Action Threshold Lactate Monthly >2 mmol/L (symptomatic) ALT/AST Baseline, Month 1,3 >5× ULN Peripheral Sensation Quarterly New numbness/tingling -
Bottle Specifications:
-
White HDPE bottle with child-resistant cap
-
“TR40” embossed on tablets
-
Desiccant canister (do not eat)
-
-
Discontinuation Criteria:
-
Confirmed lipoatrophy
-
Persistent lactate >4 mmol/L
-
Rash with mucosal involvement
-
-
Drug Interactions:
-
↓ Concentrations of methadone
-
↑ Risk of neuropathy with INH/zidovudine
-
Additional Information
| Active | Stavudine/Lamivudine/Nevirapine |
|---|---|
| Tablets | 30, 60, 90 |