- Your cart is empty
- Continue Shopping
| 🏷️ Parameter | 🔍 Details |
|---|---|
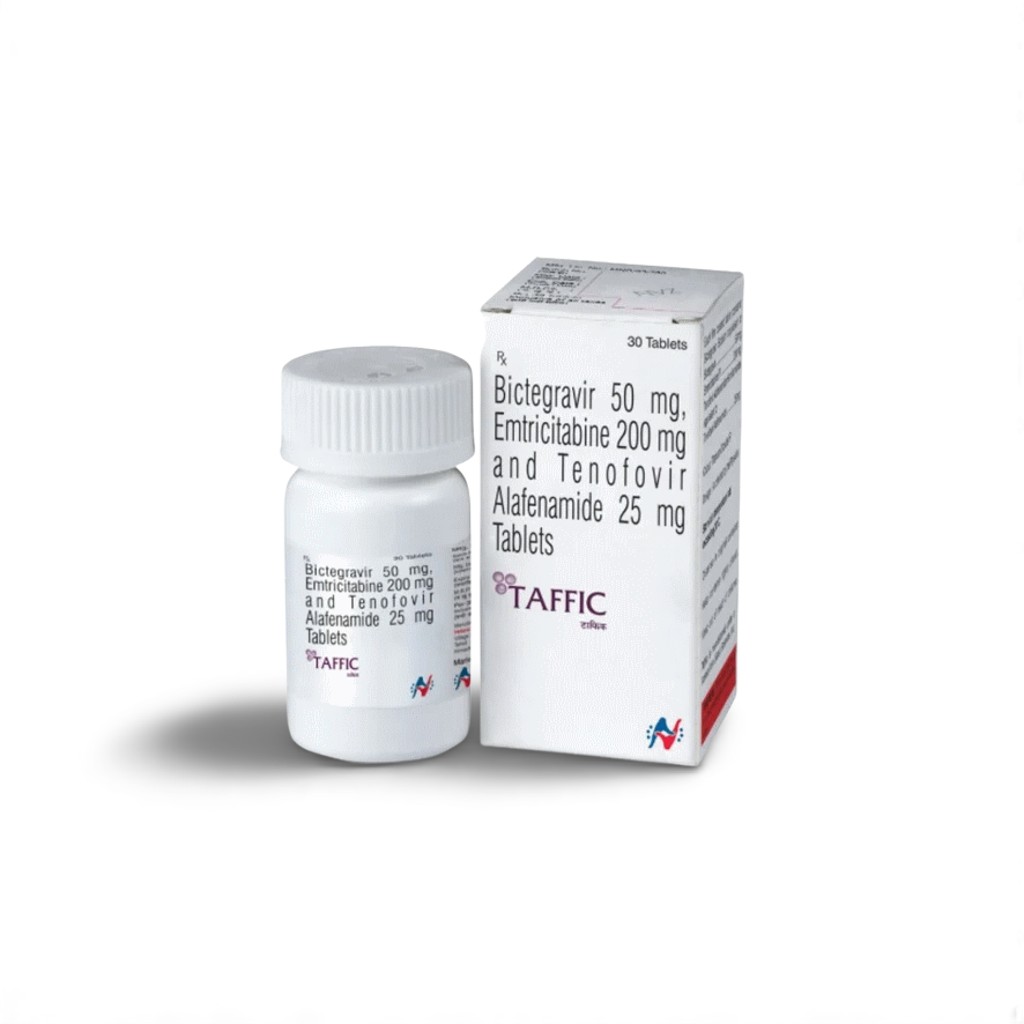
| 🧬 Active Ingredients | Bictegravir 50mg + Emtricitabine 200mg + Tenofovir Alafenamide 25mg |
| 💊 Indication | HIV-1 Infection (First-line ART for adults/adolescents ≥25kg) |
| 🇺🇸 US Brand | Biktarvy® |
| 🏭 Manufacturer | Hetero Drugs Ltd. |
| 📦 Packaging | 30 tablets in 1 HDPE bottle |
| 💪 Strength | 50mg/200mg/25mg per tablet |
| 🚚 Delivery Time | 7–14 days (Worldwide) |
🧬 Introduction to Taffic
Taffic by Hetero Drugs is a cutting-edge HIV regimen combining:
-
🧩 Bictegravir 50mg (INSTI: integrase strand transfer inhibitor)
-
🧬 Emtricitabine 200mg (NRTI: cytidine analog with dual HIV/HBV activity)
-
🛡️ Tenofovir Alafenamide 25mg (TAF: nephro-safe prodrug of tenofovir)
Approved for HIV-1 treatment in patients ≥25kg, this 30-tablet bottle offers:
-
✅ Once-daily dosing (any food/timing)
-
✅ Superior renal/bone safety vs. TDF-based regimens
-
✅ High genetic barrier to resistance
-
✅ First-line WHO-recommended regimen
-
❗ Caution with concomitant rifampicin
Hetero’s generic version provides affordable access to this gold-standard therapy.
💊 Dosage & Indications
| 💉 Indication | 🧪 Dosage | ⏱️ Frequency | 💡 Notes |
|---|---|---|---|
| Adults ≥25kg | 1 tablet | Once daily | With or without food |
| Adolescents (25-35kg) | Not recommended | – | Use pediatric formulations |
| Renal Impairment | Avoid if eGFR <30 mL/min | Contraindicated | TAF accumulation risk |
| HBV Co-infection | Never discontinue abruptly | – | Monitor for HBV flare |
⚠️ Side Effects, Storage & Warnings
-
❗ Common Side Effects:
-
Headache (10%)
-
Diarrhea (7%)
-
Nausea (6%)
-
-
🚫 Rare Serious Effects:
-
Immune reconstitution syndrome
-
Lactic acidosis (0.1%)
-
Hepatomegaly with steatosis
-
-
❄️ Storage: 15–30°C; keep bottle tightly closed (desiccant included).
-
⚠️ Critical Warnings:
-
Contraindicated with rifampicin or carbamazepine
-
Test for HBV before initiation
-
Monitor eGFR/urine protein annually
-
🌍 Global Availability & Shipping
| 🌎 Region | 📦 Shipping | 💵 Price (30-tablet bottle) | 📜 Prescription |
|---|---|---|---|
| 🇺🇸 USA | ✅ Yes | $90–$150 | Required |
| 🇬🇧 UK | ✅ Yes | £70–£120 | Required |
| 🇦🇺 Australia | ✅ Yes | AUD 130–190 | Required |
🔗 Related Indian Alternatives (Different in Same Category)
| 💊 Product Name | 🏭 Manufacturer | 🔗 Internal Link |
|---|---|---|
| Dolutranz (Dolutegravir/Tenofovir) | Mylan | dolutranz |
| Tenof-EM (Tenofovir/Emtricitabine) | Hetero | tenof-em |
| Viraday (Efavirenz/FTC/TDF) | Cipla | viraday |
| Tavin-EM (TAF/FTC) | Cipla | tavin-em |
| DoloVir (Dolutegravir/TAF/FTC) | Hetero | dolovir |
| Covihope-D (Dolutegravir/Ritonavir) | Hetero | covihope-d |
📚 External Medical References
🩺 Expert Commentary
Dr. Meena Krishnan, HIV Researcher:
“Taffic’s Bictegravir has negligible neuropsychiatric effects compared to Efavirenz-based regimens. For patients on rifampicin TB therapy, switch to [dolovir] (Dolutegravir-based). Hetero’s bottle includes oxygen absorber to maintain TAF stability in humid climates.”
📌 Disclaimer
This information is not medical advice. Use under physician supervision. Hetero’s Taffic must not be used for HBV monotherapy.
💡 Clinical Protocol:
-
HBV Co-infection Protocol:
-
Test HBV surface antigen before starting
-
If positive: Monitor LFTs monthly ×3, then quarterly
-
Never interrupt therapy (risk of fatal hepatitis flare)
-
-
Bottle Specifications:
-
White HDPE bottle with induction seal
-
Tablet ID: “H” logo + “TFC” engraving
-
Oxygen scavenger sachet (do not remove)
-
-
Drug Interactions:
Concomitant Drug Risk Action Rifampicin ↓ Bictegravir levels 75% Contraindicated Antacids ↓ Bictegravir absorption Separate by 2 hours Dabigatran ↑ Bleeding risk Monitor aPTT -
Renal Monitoring Schedule:
Parameter Frequency Action Threshold eGFR Baseline, Annual Decline >25% Urine Protein/Creatinine Annual Ratio >200 mg/g Serum Phosphate If symptoms <2.0 mg/dL
Additional Information
| active | |
|---|---|
| Tablets | 30, 60, 90 |
You must be logged in to post a review.









Reviews
There are no reviews yet.