- Your cart is empty
- Continue Shopping
| 🏷️ Parameter | 🔍 Details |
|---|---|
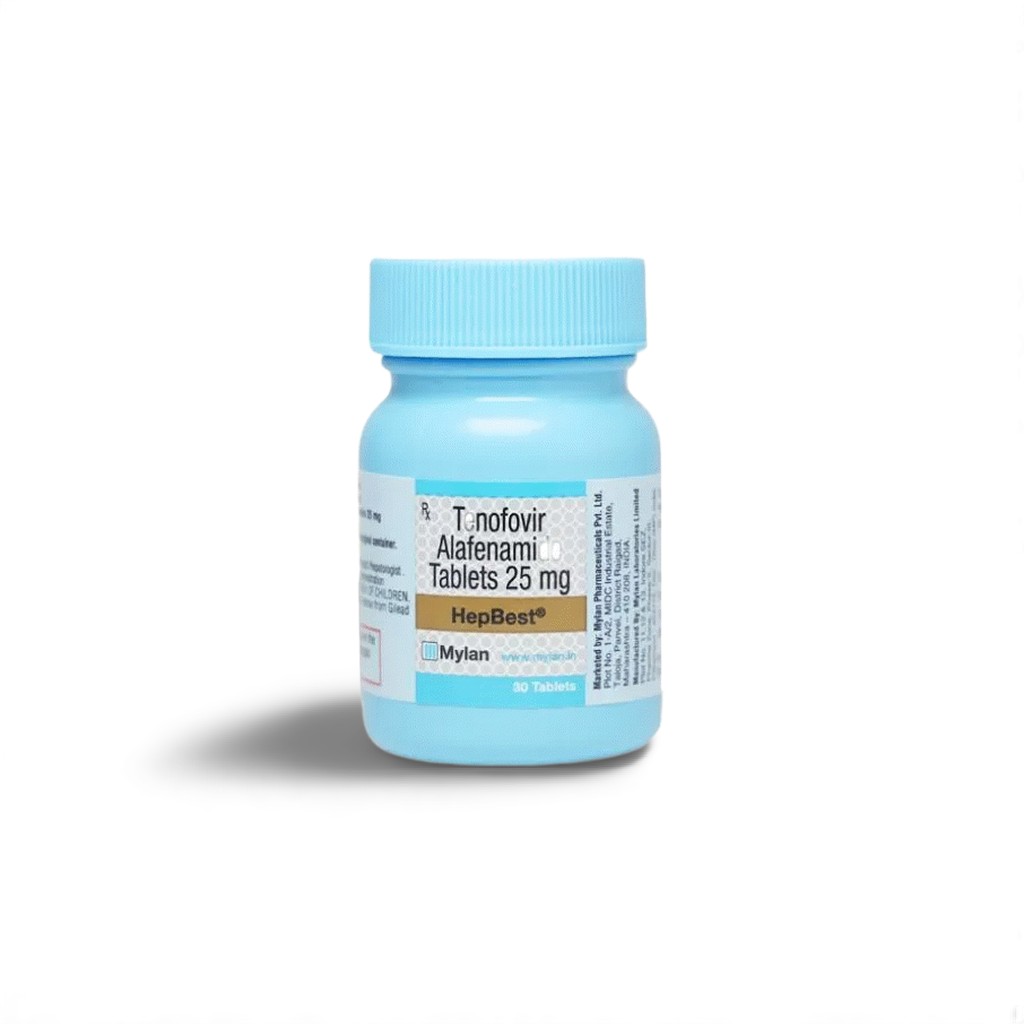
| 🧬 Active Ingredient | Tenofovir Alafenamide (TAF) |
| 💊 Indication | Chronic Hepatitis B (HBV) in adults/adolescents ≥12 years, ≥35kg |
| 🇺🇸 US Brand | Vemlidy® |
| 🏭 Manufacturer | Mylan Ltd. (FDA-approved facility) |
| 📦 Packaging | 30 tablets in 1 HDPE bottle |
| 💪 Strength | 25 mg |
| 🚚 Delivery Time | 7–14 days (Worldwide) |
⚙️ Introduction to Generic Vemlidy
This FDA-certified generic version of Vemlidy® by Hetero Drugs contains Tenofovir Alafenamide (TAF) 25mg, a next-generation antiviral for chronic hepatitis B. Key advantages:
-
🛡️ 95% lower systemic exposure vs. TDF → reduced renal/bone toxicity
-
🔬 Targeted liver delivery: Higher intracellular efficacy at lower doses
-
⚠️ Boxed warning: Severe HBV exacerbation upon discontinuation
-
✅ FDA-approved manufacturing: Hetero’s facility passed rigorous USFDA audits
Hetero’s 30-tablet bottle provides a full month’s supply. Not for HIV treatment unless part of HBV-HIV coinfection management.
💊 Dosage & Indications
| 💉 Indication | 🧪 Dosage | ⏱️ Frequency | 💡 Notes |
|---|---|---|---|
| Adults/Adolescents ≥35kg | 1 tablet (25mg) | Once daily | With food |
| Renal Impairment | No adjustment (eGFR ≥15) | – | Avoid if eGFR <15/dialysis |
| Hepatic Impairment | No adjustment | – | Safe in cirrhosis |
| HBV-HIV Coinfection | Must use with full ART | – | Prevents HIV resistance |
⚠️ Side Effects, Storage & Warnings
-
❗ Common Side Effects:
-
Headache (12%)
-
Abdominal pain (9%)
-
Fatigue (7%)
-
-
🚫 Serious Risks:
-
HBV flare (ALT elevations >10× ULN upon discontinuation)
-
Lactic acidosis (rare)
-
Immune reconstitution syndrome
-
-
❄️ Storage: 15–30°C; keep bottle tightly closed (desiccant included).
-
⚠️ Critical Warnings:
-
Contraindicated in HIV monotherapy (emerges resistance)
-
Monitor LFTs monthly for 6 months after stopping
-
Test for HIV before initiation
-
🌍 Global Availability & Shipping
| 🌎 Region | 📦 Shipping | 💵 Price (30-tablet bottle) | 📜 Prescription |
|---|---|---|---|
| 🇺🇸 USA | ✅ Yes | $120–$180 | Required |
| 🇬🇧 UK | ✅ Yes | £95–£140 | Required |
| 🇦🇺 Australia | ✅ Yes | AUD 170–220 | Required |
🔗 Related Indian Alternatives (Different in Same Category)
| 💊 Product Name | 🏭 Manufacturer | 🔗 Internal Link |
|---|---|---|
| Entavir (Entecavir) | Cipla | entavir |
| Baraclude (Entecavir) | Bristol-Myers Squibb | baraclude |
| Hepbest (Tenofovir Alafenamide) | Mylan | hepbest |
| Viread (Tenofovir DF) | Cipla | viread |
| Sebivo (Telbivudine) | Novartis | sebivo |
| Zefix (Lamivudine) | GlaxoSmithKline | zefix |
📚 External Medical References
🩺 Expert Commentary
Dr. Anil Mehta, Hepatologist:
“Hetero’s generic TAF offers 90% cost savings over branded Vemlidy with identical efficacy. For patients with eGFR <30, it’s preferable to Entecavir due to renal safety. Always confirm HIV status before prescribing – undiagnosed coinfection risks resistance development.”
📌 Disclaimer
This information is not medical advice. Use under physician supervision. Hetero’s generic Vemlidy carries a boxed warning for post-treatment hepatitis exacerbation.
💡 Clinical Protocol:
-
HBV Monitoring Schedule:
Test Frequency Action Threshold HBV DNA Baseline, Q3 months Detectable after 24 weeks ALT/AST Monthly ×6 post-D/C >10× ULN Renal Function Biannual eGFR decline >25% -
Bottle Specifications:
-
White HDPE bottle with FDA certification seal
-
Tablet ID: “HET” + “TAF25”
-
Tamper-evident cap
-
-
Drug Interactions:
Concomitant Drug Risk Action Rifampicin ↓ TAF levels 40% Avoid Antacids ↓ TAF absorption Separate by 2 hours HIV Protease Inhibitors ↑ TAF toxicity Use only with full ART -
Discontinuation Protocol:
-
Confirm undetectable HBV DNA for ≥12 months
-
Monitor LFTs monthly ×6 months
-
Restart therapy if ALT >2× ULN + detectable DNA
-
Additional Information
| Active | Tenofovir Alafenamide |
|---|---|
| Tablets | 30, 60, 90 |