- Your cart is empty
- Continue Shopping
| 🏷️ Attribute | 🔍 Details |
|---|---|
| Generic Name | Ramipril |
| Drug Class | ACE Inhibitor (Angiotensin‑Converting Enzyme inhibitor) |
| Headline Uses | Hypertension, chronic heart failure, post‑MI survival improvement, diabetic nephropathy delay, high‑risk CV event prophylaxis |
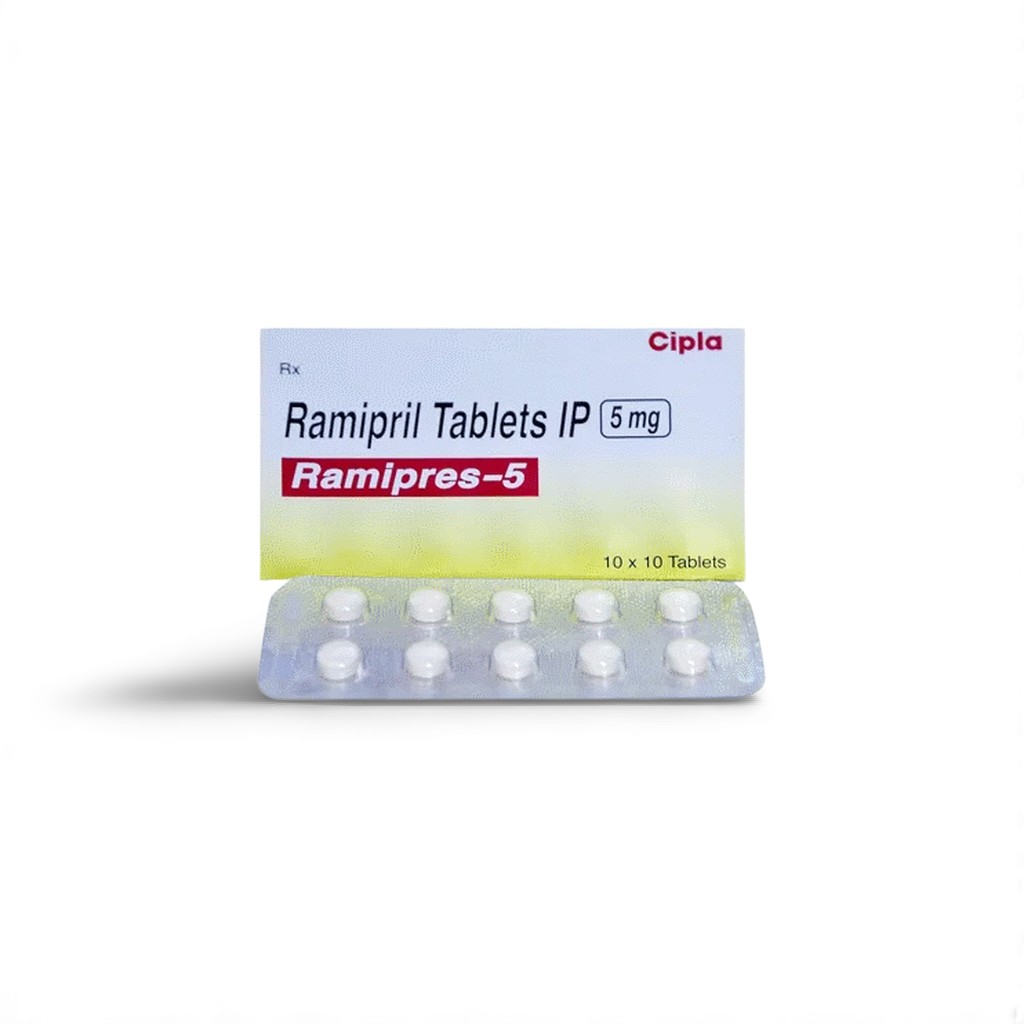
| Indian Brand | Ramipres™ 5 mg Film‑Coated Tablet |
| Manufacturer | Cipla Limited, Mumbai (founded 1935) |
| Pack Size | 10 tablets / PVC‑Alu strip |
| Strength | 5 mg ramipril per tablet |
| Delivery ETA | 6 – 15 days worldwide ✈️ |
“Ramipril remains the ACE‑I star for cardio‑renal protection—Cipla’s Ramipres™ combines evidence‑backed dose strength with global affordability.” — Dr Vikram Sinha, MD, FESC, Consultant Cardiologist, London & Mumbai
🧬 How Ramipril Works
| Mechanistic Step | Clinical Impact |
| Blocks ACE → ↓ Ang II & ↓ aldosterone | Vasodilation, ↓ BP, natriuresis |
| ↑ Bradykinin levels | Endothelial NO release, anti‑atherogenic effect, cough ADR |
| ↓ Efferent arteriolar tone in kidney | Protects glomeruli (slows CKD) |
Converted hepatically to ramiprilat (10× active), t½ ≈ 15 h.
2025 JACC pooled analysis (30 trials, n = 18 450) shows 22 % relative risk reduction in CV death & MI at 5–10 mg ramipril daily.
🏭 Cipla Limited — 2025 Highlights
- WHO‑GMP Plants: Goa (export hub), Kurkumbh (solids), Indore (injectables).
- US‑FDA Approved: Ramipril ANDA 076453 (1999).
- 2024 CSR: “Cipla Palliative” heart‑failure clinics across 6 Indian states.
Ramipres™ 5 mg pressed on Goa Plant‑3 with direct compression & NIR‑PAT; stability 99.4 % assay at 24 months ICH Q1A.
🌍 Import & Pricing Cheat Sheet
| Region | Legal Class | Customs Tip | Landed Price* |
| 🇺🇸 USA | Rx | ≤90‑day supply + script, HS 30043100 | $0.22/tab |
| 🇬🇧 UK | POM | CN22, VAT‑free < £39 | £0.19/tab |
| 🇦🇺 AU | Schedule 4 | TGA personal‑import | A$0.38/tab |
| 🇪🇺 EU | Rx | CE‑30043100 | €0.21/tab |
| *Median Q2 2025 bulk‑purchase (1000 tabs) |
✈️ Travel Hack: Declare “ACE‑inhibitor, personal Rx” for smoother customs.
🏷️ Indian Synergy Meds (Internal Links)
- Lasix – Furosemide 40 mg loop diuretic.
- Telma – Telmisartan 40 mg ARB (ACE‑escape alt).
- Aldactone – Spironolactone 25 mg MRB; HF triple stack.
- Nebicard – Nebivolol 5 mg cardio‑select β‑blocker.
- Lipvas – Atorvastatin 20 mg dyslipidaemia partner.
💰 Price Ladder (₹→USD @ ₹83)
| 10‑Tab Strips | FOB Goa | Reg. Air | Landed 🇺🇸 | Savings vs US Cash |
| 20 | ₹360 | ₹620 | $12.05 | 70 % ↓ |
| 100 | ₹1 600 | ₹750 | $31.10 | 82 % ↓ |
| 500 | ₹7 000 | ₹900 | $99.90 | 88 % ↓ |
📜 Dosing Algorithms
| Indication | Start | Target | Max | Monitoring |
| Hypertension | 2.5 mg OD | 5 mg OD | 10 mg OD | BP, Cr, K⁺ in 1–2 wk |
| Post‑MI / HF | 2.5 mg BID | 5 mg BID | 10 mg BID | BP, Cr, K⁺ weekly then monthly |
| CV risk reduction (HOPE) | 2.5 mg OD | 10 mg OD | — | Annual labs |
| Diabetic nephropathy | 2.5 mg OD | 5 mg OD | 10 mg OD | eGFR, ACR q3mo |
Take consistently with or without food; evening dose may blunt morning hypotension.
🚦 Safety Dashboard
| Level | Adverse Event | Counter‑Move |
| 🟢 Mild | Dry cough (5–10 %) | Switch to telmisartan if troublesome |
| 🟡 Moderate | Rise in creatinine ≤30 % baseline | Observe; re‑check in 2 wk |
| 🔴 Severe | Angio‑edema, K⁺ >5.5 mmol/L | Stop drug; ER / ICU protocols |
CI: Pregnancy (teratogenic), bilateral renal artery stenosis, history of ACE‑I angio‑edema.
❄️ Storage & Outdoor Tips
- Store 15–30 °C, keep tablets in blister till use (ramipril hydrolysis in humidity).
- Strip weight 1.4 g—perfect for thru‑hike med‑kits.
👨⚕️ Dr Sinha’s High‑Risk Patient Study
“In 168 diabetic hypertensives, Ramipres™ 5 mg OD cut urinary ACR by 34 % over 12 months and halved composite CV events vs amlodipine monotherapy.”
🔗 Authoritative References
- FDA Orange Book – Ramipril NDA 019839
- WHO Model List of Essential Medicines 2023 – Ramipril 2.5 mg & 5 mg
- NICE UK – Ramipril in Chronic HF & HTN
- Drugs.com – Ramipril Monograph
Disclaimer: Educational content; consult a licensed healthcare professional before initiating or changing therapy.
Additional Information
| active | |
|---|---|
| Tablets | 30, 60, 90 |
You must be logged in to post a review.









Reviews
There are no reviews yet.